Researchers at MIT are making fluorescent polymer gels that change color when they’re shaken, heated, exposed to acid, or otherwise disrupted. Given such a response, these novel materials could be effective sensors for detecting changes in structures, fluids, or the environment.
To create the gels, the researchers combine a widely used polymer with a metal that fluoresces and a chemical that can bind the two together. Mixed into a solvent, the metal and binder instantly self-assemble, grabbing the polymer molecules and pulling them together to form a gel. By using different metals, the researchers can control the physical properties of the gel as well as the color of light it emits. In a series of tests, the gels emitted a color-coded response to a variety of subtle external stimuli and later returned to their pre-stressed state and color.
Muscle mimicry
Natural organisms display some remarkable behaviors. The mussel, for example, produces strong fibers that allow it to attach tightly to boats, rocks, and other underwater surfaces. But those fibers are changeable. Pull on an attached mussel, and the stiff fibers become stretchy. Let go, and the fibers go back to their original stiff state, “self-repairing” any damage that’s occurred. In contrast, human-made materials are typically not very dynamic, and when they break, the damage is irreversible.
Niels Holten-Andersen, assistant professor in the Department of Materials Science and Engineering (DMSE) and the Doherty Professor in Ocean Utilization, has long been interested in mussel fibers and the component that’s key to their success — the metal coordination complex. This structure consists of a single metal ion (a charged particle) with several chemically bound arms, or “ligands,” radiating outward. The ligands are made of organic (carbon-containing) molecules and can attach to other molecules, enabling the complex to serve as a crosslink that binds materials together. Given that capability, the metal coordination complex plays an important role in many biological systems, including the human body, where it catalyzes enzyme-controlled reactions and binds oxygen to hemoglobin in blood.
Holten-Andersen says he’s always been fascinated by the way that nature assembles materials, putting together proteins and sugars and fatty acids in creative ways to form complex dynamic structures. “We can’t copy nature’s materials. For example, it’s difficult to synthesize proteins in the lab,” he says. “But we can see how nature builds its materials and why they work the way they do. We can then try to mimic the way nature has done it but using simple, inexpensive building blocks that we know how to make.”
To Holten-Andersen, polymer building blocks seemed like a good bet. “We know how to make simple, cheap, green polymers in large quantities,” he says. So four years ago, he decided to try making polymer gels held together by metal coordination complexes built on transition metals — a family of elements he’d frequently seen in biological settings. Initial results were promising. The polymer molecules, metals, and ligands instantly self-assembled into gels, and the mechanical properties and emitted colors of the gels depended on the transition metal used.
Adding fluorescence
Encouraged by those results, Holten-Andersen decided to try a different family of metals, the lanthanides. Like the transition metals, the lanthanides — often referred to as the rare-earth elements — provide a host of interesting and complicated behaviors. But they have one additional intriguing characteristic: They fluoresce. Shine ultraviolet light on a lanthanide, and it becomes excited and emits light at a characteristic wavelength.
“By using the lanthanides, we could still control the properties of our gels, but now we’d have light emission that would reflect any changes in those properties,” Holten-Andersen says. “With those two features intimately coupled, any time the physical properties were disturbed — say, by a change in the temperature of the nearby air or the pH of the surrounding water — the color emitted would change.” Such a polymer gel could report on its own state and serve as an excellent sensor. For example, it could be used as a coating that monitors the structural integrity of pipes, cables, and other underwater structures critical to offshore oil and gas and wind farm operations.
Testing in liquids
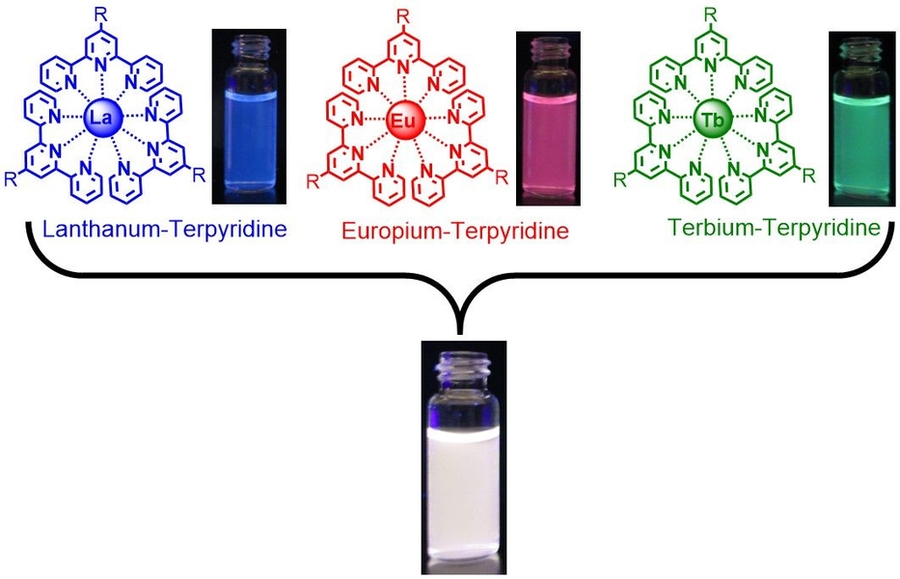
Before beginning to work with polymers, Holten-Andersen wanted to confirm — as others had shown — that mixing lanthanide ions and ligands in a solvent would produce light-emitting fluids. Accordingly, he and his team in the Laboratory for Bio-Inspired Interfaces — Pangkuan Chen, postdoc in DMSE; Qiaochu Li and Scott Grindy, both DMSE graduate students; and rising senior Rebecca Gallivan of DMSE and rising junior Caroline Liu of mechanical engineering — combined terpyridine, a commercially available ligand material, with selected lanthanides in a solvent. As Figure 1 in the slideshow above demonstrates, the mixtures produced liquids that fluoresce under ultraviolet light in the characteristic colors of the three lanthanides: blue for lanthanum, red for europium, and green for terbium.
Those results confirm that the complexes formed as expected. But a mixture emitting pure white light would make a far better sensor: It’s easier to see white light turn slightly green than it is to see green light become a little less green. To their surprise, the researchers found that producing a white-light-emitting fluid was simple. Since white light is actually a combination of many colors, they just needed to mix together their blue, red, and green light-emitting fluids. As shown in Figure 1 in the slideshow above, putting together equal parts of the three colored fluids produced a glowing white liquid.
The researchers next exposed their white-light-emitting fluid to a series of external stimuli to see if they’d get a color-coded response — and they did. For example, when they gently heated the fluid from room temperature to 55 degrees Celsius, the emitted light gradually changed color. When they let it cool down, the white light returned. The ligand and metal ions had come apart when they were heated and then reassembled when they were allowed to cool. The fluid also proved sensitive to wide-ranging changes in pH. “So we found that this simple blue-red-green approach to making a white-light-emitting system indeed leads to materials that respond to a variety of stimuli, and with that response comes a change in color,” says Holten-Andersen. The fluids might therefore serve as good sensors for detecting chemical variations within a liquid or for observing velocity gradients in fluid flow experiments — differences in flows that now must be determined indirectly by simulation.
Adding the polymer
In the next series of tests, the researchers tried incorporating their lanthanide ions and terpyridine ligands into a widely used polymer called polyethylene glycol, or PEG. At the beginning of the experiments, the polymer molecules coupled with ligands were free-floating in a solvent. “We then mixed in one of our lanthanide metals, and after some gentle shaking, the mixture changed from a fluid to a fluorescent gel,” says Holten-Andersen. The metal ions and ligands had self-assembled, linking the polymers together.
Once again, they found that gels based on different lanthanides emitted different colors, and combining them in various ratios produced shifts in color. The lead image above shows a series of gels made using europium and terbium. (It turned out they didn’t need lanthanum because the ligand itself emits blue.) The sample at the far left is all europium, therefore red; the one at the far right is all terbium, therefore blue-green; and those in between are made with various ratios of the two. Bright white luminescence appears in the second sample from the right, when the mixing ratio of terbium to europium is 96 to 4. The samples demonstrate the simplicity of designing “metallogels” with a broad spectrum of colors.
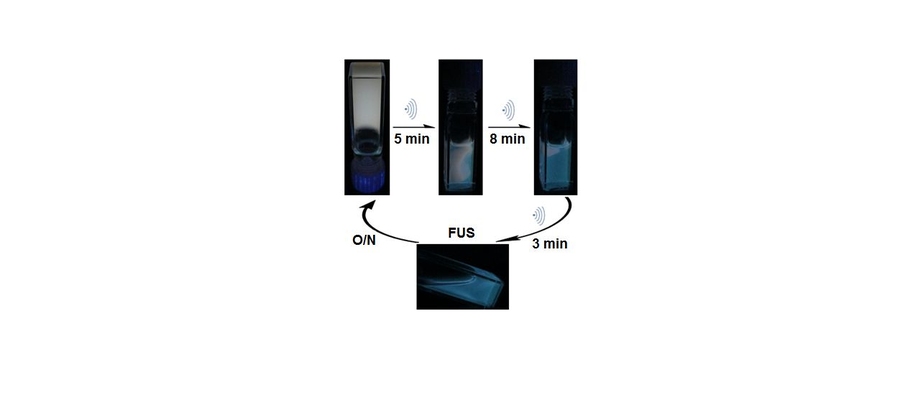
Like the fluids, the gels proved to be sensitive detectors of changes in temperature and pH. But perhaps the most dramatic response came when the gels were sonicated, that is, disrupted by exposure to high-frequency sound waves. Figure 2 above shows changes in the white-light-emitting gel during immersion in an ultrasonic bath. In the sample taken after 5 minutes, the gel is partially broken down into a fluid. The gel that remains retains its white luminescence, while the fluid gives off blue light — emitted by the now-unbound ligands. After another 11 minutes of shaking, the conversion of the sample from gel to fluid is complete, and the blue light of the ligands dominates.
And again, given time, the white-light gel reassembles. “When we let the blue fluid rest overnight, the polymers found each other again, and it turned back into a gel and made white light,” says Holten-Andersen. “That was very exciting for us because it really shows in principle that as a proof-of-concept, our approach works under these conditions. We can make a material that emits white light, reports its own failure, and then recovers. So it’s a self-reporting material that’s also self-healing.”
Looking ahead
Holten-Andersen and his team are now investigating the use of their materials as coatings that can sense structural failure as well as pH and temperature changes — a capability that will be valuable in many energy and environmental systems. Current work focuses on coatings for underwater cables used to transport electric power from offshore wind turbines to shore.
The researchers are also planning more fundamental studies. There’s a lot of interest in making materials that can change in response to various outside stimuli and then autonomously repair, returning to their original state. The availability of such self-healing materials would reduce the need to fabricate replacements for them over time.
Knowing how to build self-healing materials, however, requires knowing how those materials fail and repair in the field, and that’s difficult to study, says Holten-Andersen. He hopes their new materials may help. The chemical bonds in the metal-coordinate crosslinks have a remarkable ability to break and then re-form — and to announce that activity with changes in light emission. Guided by those light changes plus high-resolution imagery, the researchers may be able to get new insights into when, where, and how the material breaks and then comes back together.
Holten-Andersen stresses that we still have lots to learn from nature. “We’re just scratching the surface in understanding nature, given the technology we now have to look at it,” he says. For example, he believes that we’re far from finding all the metal coordination complexes that nature uses. They could occur in other natural materials with remarkable properties — perhaps in spider silk, which is tough, elastic, resilient, and one of the strongest materials known. “It’s hard to see these metals,” he says. “They appear in tiny concentrations and a single molecule at a time. But I think metal coordination complexes are much more prevalent in nature’s materials than we are currently aware of.” And coordination complexes are just one among many tricks that nature has developed over millions of years to help organisms deal with challenging environmental conditions.
This research was sponsored by the MIT Energy Initiative (MITEI) Seed Fund Program and by MIT Sea Grant via the Doherty Professorship in Ocean Utilization. Student researcher Caroline Liu received support from the Energy Undergraduate Research Opportunities Program through MITEI with funding from Lockheed Martin, a Sustaining Member of MITEI.
This article appears in the Spring 2016 issue of Energy Futures, the magazine of the MIT Energy Initiative.