Alzheimer's disease, the appalling and baffling degenerative brain illness that plagues many elderly people, may be caused by several distinct mechanisms driven by various genetic and lifestyle factors, says Li-Huei Tsai, Picower Professor of Neuroscience at MIT. To fully understand such conditions, she says, we must study the aging brain as a system rather than focusing on one or two types of ailing cells.
Neurodegenerative conditions take years to develop, partly because the brain is a highly plastic organ with many ways to adapt. “If there's one thing wrong, usually our nerve cells can figure out a way to continue to maintain the function of the brain,” Tsai says. “By the time someone shows any symptoms, the brain has already run out of any compensatory mechanism to mask the disruption. As you can imagine, this is a very systemic problem, with many things going wrong.”
Her work has led to a surprising approach to treat Alzheimer's, by increasing the strength of a certain frequency of our brainwaves. This noninvasive method has done well in early clinical trials carried out both by MIT and a startup firm co-founded by Tsai.
Director of The Picower Institute for Learning and Memory, Tsai also spearheads the “miBrain” project to create integrated multicellular models of the human brain, with all the major types of brain cells within a network of blood vessels. The miBrain models seek to offer more realistic representations of brain tissue that will allow improved testing of drug candidates — and eventually support treatments that are personalized to each patient, identified in miBrains built from their own cells. (Patients can donate skin cells that can be re-engineered to become brain cells.) Tsai is welcoming potential commercial partners to join this ambitious effort.
Catching the gamma wave
The brain bundles together nerve cells, supporting cells such as astrocytes and microglia, and blood vessels. “In Alzheimer's disease, all of these cells are disrupted,” Tsai says. “So how do you simultaneously take care of all these different systems and different cells?” Her lab has long explored stimulation methods that can engage multiple regions and cell types across the brain.
Decades ago, researchers discovered that light presented at certain frequencies in mammals can elicit nerve cells in the brain to follow along in synch, creating or strengthening brainwaves.
Tsai and MIT neuroscientist Christopher Moore examined this phenomenon in mice with a cutting-edge lab technology called optogenetics (which was originally co-developed by MIT researchers Ed Boyden and Feng Zhang when they were at Stanford University). The collaborators successfully used optogenetics to increase the power of gamma waves in rodent brains.
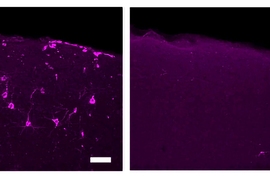
Tsai's former graduate student Hunter Iaccarino followed up to see if boosting gamma waves could produce meaningful effects in mice models of Alzheimer's disease. Working with Boyden and MIT Professor Emery Brown, Iaccarino discovered that enhancing 40-cycle-per-second gamma waves via flickering light stimulation could significantly reduce levels of the amyloid protein that is a lead indicator of Alzheimer's. The partners published these striking results in the journal Nature in 2016.
“We subsequently identified that using gamma sound stimulation also can engage nerve cells in the brain and force them to fire at the gamma frequency,” Tsai says.
The waves generated a surprising range of beneficial effects in animal models. Experiments also showed that the effect reaches key parts of the brain, such as the prefrontal cortex, where we do planning and reasoning, and the hippocampus, where we make memories.
“Today, we go cell type by cell type, system by system, to comb through all of the possible mechanisms for this effect,” she says. “If we know how it works, people will be more willing to really embrace it.”
Promise in the clinic
Early clinical trials of gamma-wave treatments have shown dramatic results.
In 2016, Tsai and Boyden were scientific co-founders of Cognito Therapeutics, a startup that has raised $93 million to commercialize gamma-wave technology. In July 2023, Cognito reported positive preliminary results for a phase 2 trial of its proprietary goggle-like device among early-stage Alzheimer's patients. Participants displayed decreased loss of brain volume and a significant slowing in functional and cognitive decline. Cognito is going forward with a phase 3 study designed to enroll 500 patients.
At MIT, Tsai and her collaborators also conducted a small-scale clinical trial on early-stage Alzheimer's subjects. Rather than giving the participants goggles, the researchers installed an LED light panel and stereo in their homes. “We reduced brain volume loss and increased connectivity,” Tsai says. This study was shut down by the pandemic, but she and her collaborators are now resuming clinical tests.
Despite employing very different devices, the Cognito and MIT trials produced similar benefits. Gamma-wave devices should be far more accessible, and safer, than the drugs available to date, Tsai suggests. Unlike the Alzheimer's drugs recently approved by the Food and Drug Administration (FDA), the therapy doesn't require highly expensive infusions or pose the risks of brain swelling and bleeding.
Modeling your whole brain with miBrain
The gamma-wave research is one thread among many in Tsai's lab aimed at understanding the entire aging brain, with its genetic complexities, and to use that understanding to personalize treatments for neurodegenerative illnesses.
“You can think of Alzheimer’s disease as like breast cancer: Depending on what genes are disrupted, people will get different therapies,” Tsai says. “I think this is probably true for other degenerative diseases, like Parkinson's.”
Her team plans to take a huge step up in modeling human brain structures with the miBrain platform, basically multicellular brain chips built with stem cell technology. (Scientists can take human skin cells and induce them to become “pluripotent” stem cells, which means they can be reprogrammed to become various brain and blood vessel cells. Those cells can then be cultured together to form a complex approximation of brain tissue.)
“This miBrain system contains all the different cell types that normally you'll see in the brain,” says Tsai. “This is essential, because the cells in the brain don't exist in isolation. They're all together and they communicate with each other through secreted factors or cell-to-cell contact, and that's a very important part of how they maintain health and functionality.”
The integrated miBrain system will empower both basic research and drug screening. “For example, the blood-brain barrier prevents many molecules from entering the brain.” she says. “We can use this in-vitro-assembled blood-brain barrier to test whether a chemical targeting a brain disease can even get into the brain.”
She points out that the FDA recently decided that it would not always require animal testing data before approving drug candidates for trial. This regulatory move should accelerate the use of in-vitro models such as the miBrain for drug testing.
Over time, Tsai hopes the miBrain will evolve into a translational platform to deliver precision medicine, enabling individualized treatments for brain illnesses. “We can reprogram your skin cells into stem cells, and then we can make a miBrain out of your cells,” she says. “If you have Parkinson's disease, we can test certain therapeutic agents and see how your miBrain responds, and then further optimize how to treat you.”
She and her MIT collaborators are now launching a miBrain center, aimed to boost both basic and translational medical research.
Scaling up this center, however, will be no easy task. “The biggest challenge is manpower, because producing all the cell types and then assembling them into a miBrain is extremely labor-intensive,” she says. “It would be very helpful if we can team up with a company and develop this together.”
MIT offers tremendous opportunities for such collaborations. “I think MIT is in the best position to lead brain disease research, because we have people who are leaders in their disciplines here, doing not just brain research but engineering and artificial intelligence,” Tsai remarks. “We really hope that we can gather people together — scientists, engineers, policymakers and economists — to figure this out. This is the future of brain research; we need people using very different approaches to work together.”