In industrial plants around the world, tiny bubbles cause big problems. Bubbles clog filters, disrupt chemical reactions, reduce throughput during biomanufacturing, and can even cause overheating in electronics and nuclear power plants.
MIT Professor Kripa Varanasi has long studied methods to reduce bubble disruption. In a new study, Varanasi, along with PhD candidate Bert Vandereydt and former postdoc Saurabh Nath, have uncovered the physics behind a promising type of debubbling membrane material that is “aerophilic” — Greek for “air-loving.” The material can be used in systems of all types, allowing anyone to optimize their machine’s performance by breaking free from bubble-borne disruptions.
“We have figured out the structure of these bubble-attracting membrane materials to allow gas to evacuate in the fastest possible manner,” says Varanasi, the senior author of the study. “Think of trying to push honey through a coffee strainer: It’s not going to go through easily, whereas water will move through, and gas will move through even more easily. But even gas will reach a throughput limit, which depends on the properties of the gas and the liquid involved. By uncovering those limits, our research allows engineers to build better membranes for their systems.”
In the paper, which appears in the journal PNAS this week, the researchers distill their findings into a graph that allows anyone to plot a few characteristics of their system — like the viscosity of their gas and the surrounding liquid — and find the best membrane to make bubble removal near-instantaneous. Using their approach, the research team demonstrated a 1,000-fold acceleration in bubble removal in a bioreactor that’s used in the pharmaceutical industry, food and beverage manufacturing, cosmetics, chemical production, and more.
The researchers say the membranes, which repel water, could be used to improve the throughput of a wide range of advanced systems whose operation has been plagued to date by bubbles.
Better bubble breakers
Companies today try everything to burst bubbles. They deploy foam breakers that physically shear them, chemicals that act as antifoaming agents, even ultrasound. Such approaches have drawbacks in tightly controlled environments like bioreactors, where chemical defoamers can be toxic to cells, while mechanical agitation can damage delicate biological materials. Similar limitations apply to other industries where contamination or physical disturbance is unacceptable. As a result, many applications that cannot tolerate chemical defoamers or mechanical intervention remain fundamentally bottlenecked by foam formation.
“Biomanufacturing has really taken off in the last 10 years,” Vandereydt says. “We’re making a lot more out of biologic systems like cells and bacteria, and our reactors have increased in throughput from 5 million cells per millimeter of solution to 100 million cells per millimeter. However, the bubble evacuation and defoaming haven’t kept up — it’s becoming a significant rate-limiting step.”
To better understand the interaction between aerophilic membranes and bubbles, the MIT researchers used MIT.nano facilities to create a series of tiny porous silicon membranes with holes ranging in size from 10 microns to 200 microns. They coated the membranes with hydrophobic silica nanoparticles.
Placing them on the surface of different liquids, the researchers released single bubbles with varying viscosity and recorded the interaction using high-speed imaging as each collided with the membranes.
“We started by trying to take a very complicated system, like foam being generated in a bioreactor, and study it in the simplest form to understand what’s happening,” Vandereydt says.
At first, the bigger the holes, the faster the bubbles disappeared. The researchers also changed the bubble gas from air to hydrogen, which has half the viscosity, and found the speed of bubble destruction doubled.
But after about a 1,000-fold acceleration in bubble destruction, the researchers hit a wall no matter how big the membrane holes were. They had run up against a different physical limit to investigate.
The researchers then tried changing the viscosity of their liquid, from water to something closer to honey. They found viscosity only plays a role in the speed of bubble destruction when the liquid is 200 times the viscosity of liquid. Further experiments revealed the biggest factor for slowing bubble evacuation was inertial resistance in the liquid.
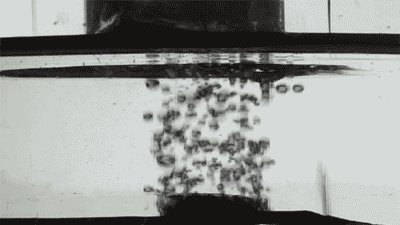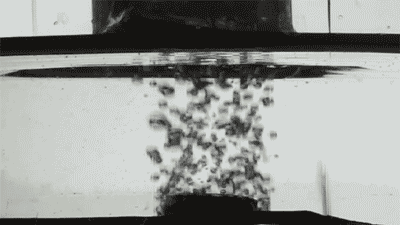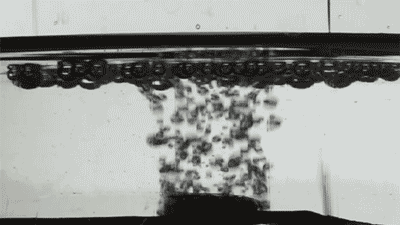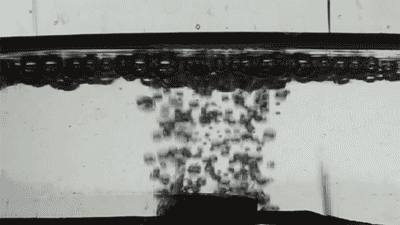
Credit: Courtesy of the researchers
“Through experimentation, we showed there are three different limits [to the speed of bubble destruction],” Vandereydt says. “There is the viscous limit of the gas in a low-viscosity, low-permeability setup. Then there’s the viscous resistance of the liquid in the high-permeability, high-viscosity regime. Then we have the inertial limit of the liquid.”
The team used a bioreactor to experimentally validate their findings and charted them in a map that engineers can use to enter the characteristics of their system and find both the best membrane for their situation and the biggest factor slowing bubble evacuation.
The science of bubbles
The research should be useful for anyone trying to accelerate the destruction of bubbles in their industrial device, but it also improves our understanding of the physics underpinning bubble dynamics.
“We have identified three different throughput limits, and the physics behind those limits, and we have reduced it to very simple laws,” Nath explains. “How fast you can go is first dictated between surface tension and inertia. But you may also hit a different limit, where the pores are extremely small, so the gas finds it difficult to move through them. In that case, the viscosity of the gas is meaningful. But you may also have a bubble which was originally in something like honey, which means it’s not enough the gas is moving, the liquid also must refill the space behind it. No matter what your conditions are, you will be switching between these three limits.”
Varanasi says health care companies, chemical manufacturers, and even breweries have expressed interest in the work. His team plans to commercially develop the membranes for industrial use.
“These physical insights allowed us to design membranes that, quite surprisingly, evacuate bubbles even faster than a free liquid-gas interface,” says Varanasi.
The researchers’ design map could also be used to model natural systems and even liquid-liquid systems, which could be used to create membranes that remove oil spills from water or help efficiently extract hydrogen from water-splitting electrodes. Ultimately the biggest beneficiaries of the findings will be companies grappling with bubbles.
“Though small, bubbles quietly dictate the performance limits of many advanced technologies,” says Varanasi. “Our results provide a way to eliminate that bottleneck and unlock entirely new levels of performance across industries. These membranes can be readily retrofitted into existing systems, and our framework allows them to be rapidly designed and optimized for specific applications. We’re excited to work with industry to translate these insights into impact.”
The work was supported, in part, by MIT Lincoln Laboratory and used MIT.nano facilities.