Many of the most promising new pharmaceuticals coming along in the drug development pathway are hydrophobic by nature — that is, they repel water, and are thus hard to dissolve in order to make them available to the body. But now, researchers at MIT have found a more efficient way of processing and delivering these drugs that could make them far more effective.
The new method, which involves initially processing the drugs in a liquid solution rather than in solid form, is reported in a paper in the Dec. 15 print issue of the journal Advanced Healthcare Materials, written by MIT graduate student Lucas Attia, recent graduate Liang-Hsun Chen PhD ’22, and professor of chemical engineering Patrick Doyle.
Currently, much drug processing is done through a long series of sequential steps, Doyle explains. “We think we can streamline the process, but also get better products, by combining these steps and leveraging our understanding of soft matter and self-assembly processes,” he says.
Attia adds that “a lot of small-molecule active ingredients are hydrophobic, so they don’t like being in water and they have very poor dissolution in water, which leads to their poor bioavailability.” Giving such drugs orally, which patients prefer over injections, presents real challenges in getting the material into the patient’s bloodstream. Up to 90 percent of the candidate drug molecules being developed by pharmaceutical companies actually are hydrophobic, he says, “so this is relevant to a large class of potential drug molecules.”
Another advantage of the new process, he says, is that it should make it easier to combine multiple different drugs in a single pill. “For different types of diseases where you’re taking multiple drugs at the same time, this kind of product can be very important in improving patient compliance,” he adds — only having to take one pill instead of a handful makes it much more likely that patients will keep up with their medications. “That’s actually a big issue with these chronic illnesses where patients are on very challenging pill regimes, so combination products have been shown to help a lot.”
One key to the new process is the use of a hydrogel — a sort of sponge-like gel material that can retain water and hold molecules in place. Present processes for making hydrophobic materials more bioavailable involve mechanically grinding the crystals down to smaller size, which makes them dissolve more readily, but this process adds time and expense to the manufacturing process, provides little control over the size distribution of the particles, and can actually damage some more delicate drug molecules.
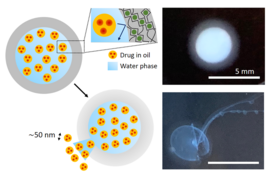
Instead, the new process involves dissolving the drug in a carrier solution, then generating tiny nanodroplets of this carrier dispersed throughout a polymer solution — a material called a nanoemulsion. Then, this nanoemulsion is squeezed through a syringe and gelled into a hydrogel. The hydrogel holds the droplets in place as the carrier evaporates, leaving behind drug nanocrystals. This approach allows precise control over the final crystal size. The hydrogel, by keeping the droplets in place as they dry, prevents them from simply merging together to form lumpy agglomerations of different sizes. Without the hydrogel the droplets would merge randomly, and “you’d get a mess,” Doyle says. Instead, the new process leaves a batch of perfectly uniform nanoparticles. “That’s a very unique, novel way that our group has invented, to do this sort of crystallization and maintain the nano size,” he says.
The new process yields a two-part package: a core, which contains the active molecules, surrounded by a shell, also made of hydrogel, which can control the timing between ingestion of the pill and the release of its contents into the body.
“We showed that we can get very precise control over the drug release, both in terms of delay and rate,” says Doyle, who is the Robert T. Haslam Professor of Chemical Engineering and Singapore Research Professor. For example, if a drug is targeting disease in the lower intestine or colon, “we can control how long until the drug release starts, and then we also get very fast release once it begins.” Drugs formulated the conventional way with mechanical nanomilling, he says, “would have a slow drug release.”
This process, Attia says, “is the first approach that can form core-shell composite particles and structure drugs in distinct polymeric layers in a single processing step.”
The next steps in developing the process will be to test the system on a wide variety of drug molecules, beyond the two representative examples that were tested so far, Doyle says. Although they have reason to believe the process is generalizable, he says, “the proof is in the pudding — having the data in hand.”
The dripping process they use, he says, “can be scalable, but there’s a lot of details to be worked out.” But because all of the materials they are working with have been chosen as ones that are already recognized as safe for medical use, the approval process should be straightforward, he says. “It could be implemented in a few years. … We’re not worrying about all those typical safety hurdles that I think other novel formulations have to go through, which can be very expensive.”
The work received support from the U.S. Department of Energy.