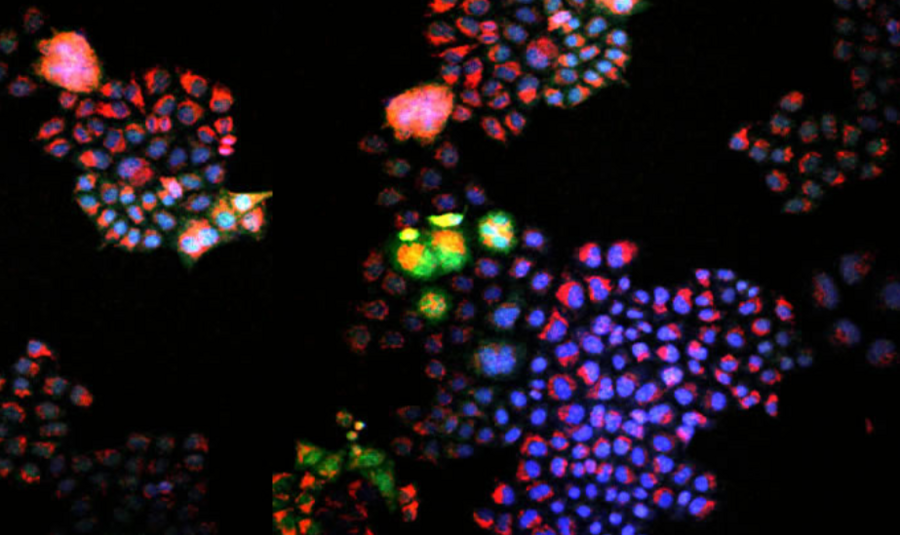
MIT chemical engineers have developed a way of swiftly screening compounds to determine their therapeutic potential for certain kinds of cancers. With a genetically engineered sensor and high-throughput technology, their method probes for changes in cellular concentrations of hydrogen peroxide (H2O2), a specialized molecule known as an oxidant.
“The regulatory pathways of some tumors depend on elevated levels of H2O2,” says Hadley Sikes, associate professor and Esther and Harold E. Edgerton Career Development Professor in the Department of Chemical Engineering. “But further increases in concentrations of this oxidant can lead to programmed cell death.” In the researchers’ screens of 600 small-molecule compounds, they were able to identify those that selectively boosted H2O2.
Other research efforts have used probes that respond indiscriminately to different kinds of oxidants, making it difficult to determine precisely which compounds make the greatest impact on these specialized molecules. The MIT screen is the first to zero in on a single oxidant. This enabled the team to characterize the cellular responses to potential drugs and to demonstrate that some of these compounds activated H2O2-mediated toxicity in susceptible cancer cell lines.
Their research appears in Cell Chemical Biology. Yining Hao SM ’18 and Troy F. Langford SM ’15 PhD ’18 are first co-authors. The other contributors are Sun Jin Moon, a graduate student in chemical engineering, Kristen A. Eller ’16, who worked on the project while an undergraduate, and Sikes.
“Our work helps pave the way for highly targeted, oxidant-based chemotherapeutics,” says Hao, who is completing her doctorate in chemical engineering. “These studies move us in the right direction for effectively utilizing drugs to treat different patients — the idea behind personalized medicine,” adds Langford, now a biotechnology associate for Cowen, Inc.
Programmed cell death
Hydrogen peroxide belongs to a family of molecules called reactive oxygen species (ROS), which are involved in metabolizing oxygen. “They’re known as 'Jekyll-and-Hyde' molecules,” says Sikes. “They are part of all the things we need to live — taking oxygen from the air, reducing it to water, generating energy for the cells — but uncontrolled concentrations of ROS for too long can have negative effects, such as interfering with the signaling pathways inside cells.”
When genetic mutations kick-start cancers, sometimes oxidants such as hydrogen peroxide increase dramatically, throwing cell functions out of gear. As levels of hydrogen peroxide rise, cancer cells unleash antioxidants to keep them in check. It is a difficult metabolic balance to maintain, and it is this weakness that researchers hope to exploit as they seek new cancer therapies.
“The idea is, if we selectively boost hydrogen peroxide, these stressed cancer cells will die first,” says Hao. “We are looking for molecular vulnerabilities that will have a greater impact on cancer than on the healthy tissues that surround it,” adds Sikes.
Drugs have emerged for the anti-cancer arsenal that aim to act on these hydrogen peroxide mechanisms, either by directly elevating cellular levels of the oxidant, or by undermining antioxidant systems. But they do not uniformly deliver. Without a sure-fire method for detecting hydrogen peroxide in cancer cells before and after drug treatment, precision therapeutics remains out of reach.
The biosensor Langford and Sikes devised in 2018 addressed this problem. It uses an enzyme called peroxiredoxin-2, which can register changes in hydrogen peroxide levels. Langford engineered the sensor so that when it reacts with hydrogen peroxide, it fluoresces.
“We wanted to use this sensor in a practical manner, and we thought: 'What better way to do that than a high-throughput screen, using a library of anti-cancer compounds right next door at the Koch Institute for Integrative Cancer Research?'” says Langford. “We took these small molecules from their collection and systematically added each one to cancer cells that contained our sensor.”
Sikes made the deliberate decision to take compounds that were “already FDA-approved and safe in humans,” she says, including some that had previously been investigated as anti-cancer drugs. The question was which, if any, could be effective in elevating concentrations of hydrogen peroxide in the human cancer cell lines the team had assembled.
Lighting up
As they ran their screens, researchers looked for the red fluorescing of the probe that signaled an increase in hydrogen peroxide levels after the drugs interacted with the cells. There were indeed such hits, but in data analysis, Hao found that “a lot of these signals were weirdly high, outside of the sensor’s range.”
The researchers ran a second round, to make sure the signals actually reflected hydrogen peroxide level changes. After running through the library of drug candidates, the researchers not only pinpointed compounds that modulated hydrogen peroxide in specific cancer cells, but also linked some of those compounds to the death of cells.
There were surprises. One drug, SMER3, an antifungal, ramped up hydrogen peroxide levels. “This was the most exciting one we came across,” says Sikes. “It kills yeast and it turned out to be effective killing a subset of cancers.”
Another headline from their study: A major anti-cancer drug with a reputation for boosting oxidant levels was a bust in their screen. “Cisplatin did not induce hydrogen peroxide,” says Sikes. “Maybe it induces other oxidants, but not this one that drives death response pathways in a subset of cancers.”
Their work has already catalyzed new enterprises. Sikes is partnering with a Boston clinician who specializes in cancers known to be vulnerable to ROS defects, such as colon cancer. As part of his dissertation research, Langford tested SMER3 on colon cancer cells. “It kills certain cell lines very efficiently,” Hao says, “and we’d like to understand it better, to learn if it’s safe, and what cell pathways it targets exactly.”
Next steps involve moving on to animal models bearing patient-derived cancers, says Sikes, and eventually on to patient populations.
Beyond hydrogen peroxide, Sikes envisions taking up other molecules that fulfill critical functions in cells, and that might also serve as potent anti-cancer targets. “There are reactive nitrogen and sulfur species that are also likely equally important and worthy of exploration.”
Support for this research came from the Haas Family Fellowship in Chemical Engineering, the MIT Advanced Undergraduate Research Opportunities Program, and the Esther and Harold E. Edgerton endowed professorship.