After its emergency meeting on Zika virus two weeks ago, World Health Organization (WHO) Director-General Margaret Chan wrote in an official statement, “the Committee advised that the recent cluster of microcephaly cases and other neurological disorders reported in Brazil, following a similar cluster in French Polynesia in 2014, constitutes an ‘extraordinary event’ and a public health threat to other parts of the world.”
Given these stakes, WHO declared Zika a “global public health emergency." The designation highlights immediate goals, such as “development of new diagnostics . . . prioritized to facilitate surveillance and control measures,” and longer-term measures, such as “appropriate research and development efforts . . . intensified for Zika virus vaccines, therapeutics and diagnostics.”
MIT’s Institute of Medical Engineering and Science (IMES) is on the front lines of this research.
Rapid diagnostics
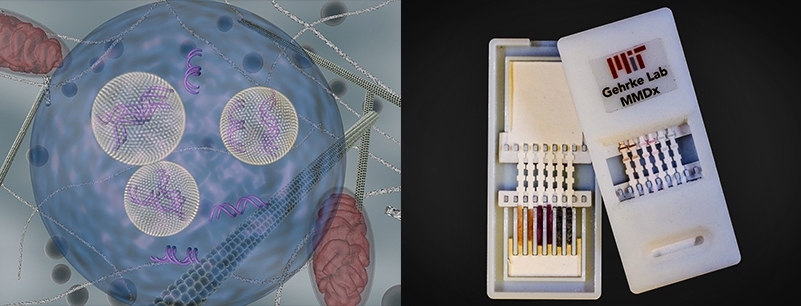
Led by Professor Lee Gehrke, the MIT Gehrke Lab is at the vanguard of disease diagnostics. In recent years, lab members have developed a successful, noncross-reacting diagnostic test for Dengue virus, a close relative to Zika, as well as one for Ebola.
“The Gehrke team is currently focused on developing sensitive, low-cost, rapid diagnostics to detect Zika, giving a result in 10 to 15 minutes, as well as on basic research needed to characterize Zika virus infections in advance of therapeutics and vaccine development,” says Gehrke, the Hermann von Helmholtz Professor in IMES and Professor of Microbiology and Immunobiology at Harvard Medical School. “Detection is needed from an epidemiological perspective . . . [and] to improve patient care. By detecting specific pathogens, a tailored treatment plan can be developed.”
The threat is compounded by several factors, Gherke says: “Because there is little immunity against Zika virus infections in most parts of the world, Zika has the potential to spread anywhere that the mosquito vectors (Aedes aegypti and Aedes albopictus) are found.
“Further, there are few available research reagents that can be used for scientific experimentation and study. Yet the development and characterization of reagents needed to detect Zika virus is underway at a rapid pace now, [and] we hope that the Zika test will be ready for field-testing in late spring or early summer 2016.
Gehrke adds that his team has been indespensible in the work: “MIT graduate student Helena de Puig is developing nanoparticle surface chemistries to increase the sensitivity of the tests, and undergraduate Luis Mora is taking time from his work on the MIT Solar Car to precision-mold the cartridges that hold the device,” he says.
Because Zika disease is often nonsymptomatic, several public health concerns are presently at play, Gehrke says. “There might be some danger from nonsymptomatic individuals serving as virus sources for mosquitoes, who could in turn infect other people,” he explains. “Once an individual has been infected, it’s reasonable to predict that they will have immunity, but no one knows the long term effects of Zika infections.”
For this reason, Gehrke’s lab wants to provide health workers with a rapid test to detect who has Zika and does not, as well as who’s had it in the past. U.S. Centers for Disease Control Director Tom Friedan wrote for CNN: “Diagnosing prior infection with Zika is much more challenging. . . . This is a priority, and we are working to do in weeks what would usually take months or years.”
“Identifying individuals who have been infected in the past is done by identifying anti-Zika IgM and IgG antibodies,” Gehrke says. “Advanced technology is available for examining the portfolio of pathogens that have infected an individual over time. However, as a practical matter with readily available reagents, the IgG and IgM tests can be non-specific. Our Dengue Fever device can identify IgG and IgM in patient serum, and we will develop the same capacity for Zika. Crossover interactions with nonspecific antigens are, however, a consideration.”
Developing a messaging system to prevent an epidemic
Since reporting outbreaks is crucial to public health, technology is also being implemented to make diagnostics into messengers.
“The detection technology is being linked to low-cost communication and data technologies that will analyze the test results and transmit them to personal care physicians and public health officials,” Gehrke explains. “[IMES researcher] Jose Gomez Marquez is adapting a 5-dollar Raspberry Pi single-board computer to invent a very low-cost reader that will quantify the rapid test data. More research is needed to determine how that data can be transmitted and stored to protect privacy while improving communication with physicians and public health officials.”
Additionally, the Gehrke Lab wants to help officials to “develop a plan for a ‘smart’ mosquito trap that identifies mosquito species as they enter and transmits the data to a home base. We are also exploring technologies to screen mosquitoes directly for the presence of viruses, in an effort to provide advance warning of an impending epidemic.”
Ultimately, the sudden, intensive focus on Zika could help the fight against other viruses, as well. Gehrke’s own lab is focused on detecting multiple RNA viruses, zeroing in on processes from innate immune signaling to gene expression.
“Because the genomes and proteins of the flaviviruses (including Zika) are related, it is quite possible that studying Zika and comparing its replication strategies and pathogenesis to other viruses will be very informative,” Gehrke says.
New approach to vaccines
Daniel Anderson, an associate professor of chemical engineering and member of IMES, is working on a new type of vaccine: a customized, on-hand, single-dose RNA nanoparticle vaccine — that may have potential to fight the spread of Zika virus.
“We have developed a nanoparticle vaccine that can be rapidly tuned for different diseases, including viruses. The speed of development allows us to make new vaccines in only seven days, allowing the potential to deal with sudden outbreaks, like Zika,” Anderson says. “Using a single injection, these nanoparticles deliver RNA, carrying instructions that ultimately help train the patient’s immune system to fight off infections.”
The team at the Anderson Lab has successfully developed customized vaccines for all three classes of National Institute of Allergy and Infectious Disease’s “priority pathogens,” including Ebola virus (class A), H1N1 Influenza (class B), and microorganism parasite Toxoplasma gondii (class C), generating fully protective immunity in animals.
“We have already started using the same process for Zika,” says postdoc Omar F. Khan, who works closely with Anderson at the Koch Institute for Integrative Cancer Research at MIT. “It’s possible that some of the antigens found in Zika are also present in other arboviruses [viruses caused by mosquito or tick bites], which broadens the vaccine’s applicability.”
In fact, by designing the synthetic nanoparticle to “simultaneously deliver many different and unique RNAs,” the Anderson Lab’s vaccine innovation has the ability to target multiple diseases, reducing the burden on health care workers addressing large populations.
“These can be multiple antigens from the same virus or different antigens from other viruses. We call this multiplexing,” Khan says. “In this way, it’s possible to make a pan-arbovirus vaccine that can simultaneously train the immune system to target [related viruses]. We are already doing this type of multiplexing for filoviruses. Our pan-filovirus nanoparticle vaccine is designed to simultaneously target Zaire Ebola, Sudan Ebola, and Marburg virus, and is currently being validated by our collaborators at the United States Army Medical Research Institute for Infectious Diseases.”
The speed, cost, and on-demand, local production potential of a nanoparticle vaccine for Zika — or Zika combined with other arboviruses — makes it particularly compelling.
“Once the unique Zika antigens have been identified by virology experts, it will take us about seven days to generate the Zika-specific RNA instructions and build the nanoparticle vaccines,” Anderson says. “We are working with labs and government agencies that study the Zika virus to validate the potential in preventing this disease.”
The nanoparticle vaccine is, according to Anderson’s presentations, the “first and only nonviral replicon delivery system that has achieved protective immunity in lethal exposure experiments . . . [with] no side effects [and] no risk of anti-vector immunity” — in other words, no risk of being attacked by the patient’s immune system. And based on its success preventing other viruses, influenza, parasites, and even some cancers in animals, it offers promising potential as a formidable foe to Zika in the future, if clinical trials are prioritized.