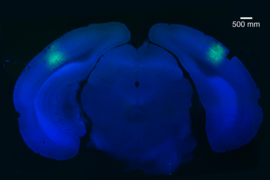
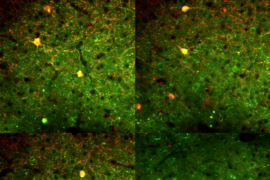
Researchers at MIT’s Picower Institute for Learning and Memory have identified a master genetic regulator that could account for faulty brain functions that contribute to schizophrenia.
The work, reported May 25 in the advance online publication of Nature Neuroscience, may one day lead to new strategies for treating schizophrenia and other diseases caused by malfunctioning synapses, the connections among neurons, said co-author Li-Huei Tsai, director of the Picower Institute and the Picower Professor of Neuroscience at MIT.
Messenger RNA (mRNA) translates DNA’s genetic information into codes for proteins. A recently discovered class of noncoding RNA molecules, short stretches of RNA called microRNAs (miRNAs) has been shown to interact with mRNA and block protein production.
More than 3,000 miRNAs have been found in the human genome. A single miRNA can regulate the activity of hundreds of genes, thus impacting entire cellular networks.
Genetic variations in the gene encoding a particular microRNA — miRNA-137 — have been linked with an increased risk for schizophrenia. While schizophrenia is tied to multiple genes, “a more complete picture of miRNAs may improve our understanding of the molecular mechanisms underlying schizophrenia,” Tsai says.
Exactly how genetic variations impact miRNA-137 levels — and effects of miRNA-137 in the brain — is unclear. “It’s important to understand how abnormal levels of miRNA-137 impact cellular function,” says co-author Sandra Siegert, a postdoc in the Picower Institute.
Siegert found that in reprogrammed neurons from human fibroblasts of diseased patients, microRNA-137 was increased. This was, in turn, tied to presynaptic defects and impaired synaptic plasticity and cognitive dysfunction. This was a surprise, she says, because although schizophrenia is considered “a disease of the synapse,” nobody had ever shown that microRNAs could influence the mechanism through which neurotransmitters are released at the synapse.
“This work demonstrates that miRNAs directly impact synaptic proteins,” Tsai says. “This shows a master regulator role for MiRNA-137 as an exciting common regulator of presynaptic function.” The study proves that “just by down-regulating miRNA-137, we were able to improve synaptic transmission in neurons,” Siegert says. “This is very promising, and shows the beneficial effects” of manipulating miRNA-137.
MiRNAs also have been found to be a gateway for environmental factors’ influence on cellular mechanisms. For instance, drug abuse or stress can alter miRNA levels. “If we knew which environmental factors affected MiRNA-137, there might be a simple approach to tweaking it. If genetic testing showed an individual was a carrier of the risk-related variation of the MiRNA-137 gene, avoiding an unhealthy lifestyle or drugs in a critical phase of brain development might prevent disease onset,” Siegert says.
This work is supported by the Simons Center for the Social Brain and the National Institutes of Health.