A class of cancer drugs called protein kinase inhibitors is one of the most effective treatments for melanoma. However, in many cases, tumors eventually become resistant to the drugs and cause a relapse in the patient.
A new study from MIT suggests that combining kinase inhibitors with experimental drugs known as ribonucleases could lead to better results. In tests with human cancer cells, the researchers found that the two drugs given together kill cells much more effectively than either drug does on its own. The combination could also help to prevent tumors from developing drug resistance, says Ronald Raines, the Firmenich Professor of Chemistry at MIT.
“We discovered that this ribonuclease drug could be paired favorably with other cancer chemotherapeutic agents, and not only that, the pairing made logical sense in terms of the underlying biochemistry,” Raines says.
Raines is the senior author of the study, which appears in the Dec. 3 issue of Molecular Cancer Therapeutics and was posted in the journal’s “online first” section on Nov. 20. Trish Hoang, a former graduate student at the University of Wisconsin at Madison, is the lead author of the study.
Unexpected link
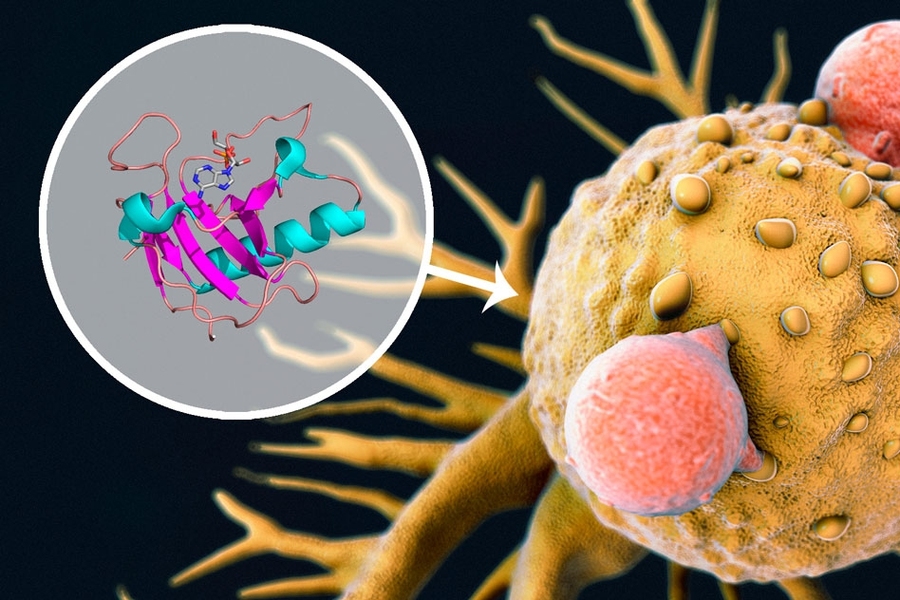
Ribonucleases are enzymes produced by all human cells that break down RNA molecules. They degrade cellular RNA that is no longer needed, and they help to defend against viral RNA. Because of ribonucleases’ ability to kill cells by damaging their RNA, Raines has been working on developing these enzymes as cancer drugs for about two decades.
His lab has also been studying the protein that has evolved to help cells defend against ribonucleases, which can be very destructive if unchecked. This protein, called ribonuclease inhibitor, binds to ribonucleases with a half-life of at least three months — the strongest naturally occurring protein-binding interaction ever recorded. “That means that should ribonuclease invade cells, there is an unbelievable defense system,” Raines says.
To create a ribonuclease drug for testing, the researchers modified it so that ribonuclease inhibitors don’t bind as tightly — the half-life for the interaction is only a few seconds. One version of this drug is now in a phase 1 clinical trial, where it has stabilized the disease in about 20 percent of patients.
In the new study, the researchers found an unexpected link between ribonucleases and enzymes called protein kinases (the targets of protein kinase inhibitors), which led them to discover that the two drugs can kill cancer cells much better when used together than either one can alone.
The discovery came about when Hoang decided to try to produce the ribonuclease inhibitor protein in human cells instead of in E. coli, which Raines’ lab normally uses to produce the protein. She found that the human-cell-produced version, though identical in amino acid sequence to the protein produced by bacteria, bound to ribonucleases 100 times more strongly. This boosted the half-life of the interaction from months to decades — a protein-binding strength previously unheard of.
The researchers hypothesized that human cells were somehow modifying the inhibitor in a way that made it bind more tightly. Their studies revealed that, indeed, the inhibitor produced by human cells had phosphate groups added to it. This “phosphorylation” made the inhibitor bind much more strongly than anyone had previously suspected.
The researchers also discovered that phosphorylation was being carried out by protein kinases that are part of a cell signaling pathway called ERK. This pathway, which controls how cells respond to growth factors, is often overactive in cancer cells. The protein kinase inhibitors trametinib and dabrafenib, used to treat melanoma, can shut off the ERK pathway.
“This was a fortuitous intersection of two different strategies, because we reasoned that if we could use these drugs to deter the phosphorylation of ribonuclease inhibitor, then we could make the ribonucleases more potent at killing cancer cells,” Raines says.
Combating resistance
Tests of human melanoma cells supported this idea. The combination of a kinase inhibitor plus a ribonuclease was much deadlier to cancer cells, and the drugs were effective at lower concentrations. The kinase inhibitor prevented the ribonuclease inhibitor from being phosphorylated, making it weaker and allowing the ribonuclease more freedom to perform its function and destroy RNA.
If the same holds true in human patients, this approach could lead to reduced side effects and a lower chance of tumor cells becoming drug-resistant, Raines says. The researchers now hope to test this drug combination in mice, as a step toward testing the combination in clinical trials.
“We’re hoping that we can explore relationships with some of the many pharmaceutical companies that develop ERK pathway inhibitors, to team up and use our ribonuclease drug in concert with kinase inhibitors,” Raines says.
The researchers have also engineered mice that do not produce ribonucleases, which they plan to use to further study the biological functions of these enzymes.
The research was funded by the National Institutes of Health.