All growth and reproduction relies on a cell’s ability to replicate its chromosomes and produce accurate copies of itself. Every step of this process takes place within that cell.
Based on this observation, scientists have studied the replication and segregation of chromosomes as a phenomenon exclusively internal to the cell. They traditionally rely on warm nutritional cultures that promote growth but bear little resemblance to the cell’s external surroundings while in its natural environment.
New research by a group of MIT biologists reveals that this long-held assumption is incorrect. In a paper published this week, they describe how some types of cells rely on signals from surrounding tissue in order to maintain chromosome stability and segregate accurately.
Kristin Knouse, a fellow at the Whitehead Institute, is the lead author of the paper, which was published online in the journal Cell on Aug. 23. Angelika Amon, the Kathleen and Curtis Marble Professor in Cancer Research in the Department of Biology and a member of the Koch Institute for Integrative Cancer Research, is the senior author.
“The main takeaway from this paper is that we must study cells in their native tissues to really understand their biology,” Amon says. “Results obtained from cell lines that have evolved to divide on plastic dishes do not paint the whole picture.”
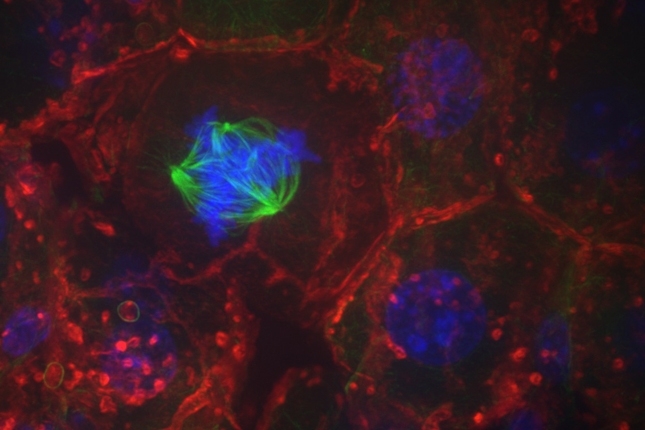
When cells replicate, the newly duplicated chromosomes line up within the cell and cellular structures pull one copy to each side. The cell then divides down the middle, separating one copy of each chromosome into each new daughter cell.
At least, that’s how it’s supposed to work. In reality, there are sometimes errors in the process of separating chromosomes into daughter cells, known as chromosome mis-segregation. Some errors simply result in damage to the DNA. Other errors can result in the chromosomes being unevenly divided between daughter cells, a condition called aneuploidy.
These errors are almost always harmful to cell development and can be fatal. In developing embryos, aneuploidy can cause miscarriages or developmental disorders such as Down syndrome. In adults, chromosome instability is seen in a large number of cancers.
To study these errors, scientists have historically removed cells from their surrounding tissue and placed them into easily controlled plastic cultures.
“Chromosome segregation has been studied in a dish for decades,” Knouse says. “I think the assumption was … a cell would segregate chromosomes the same way in a dish as it would in a tissue because everything was happening inside the cell.”
However, in previous work, Knouse had found that reported rates for aneuploidy in cells grown in cultures was much higher than the rates she found in cells that had grown within their native tissue. This prompted her and her colleagues to investigate whether the surroundings of a cell influence the accuracy with which that cell divided.
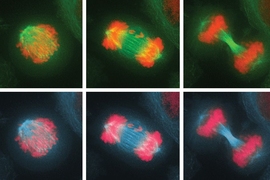
To answer this question, they compared mis-segregation rates between five different cell types in native and non-native environments.
But not all cells’ native environments are the same. Some cells, like those that form skin, grow in a very structured context, where they always have neighbors and defined directions for growth. Other cells, however, like cells in the blood, have greater independence, with little interaction with the surrounding tissue.
In the new study, the researchers observed that cells that grew in structured environments in their native tissues divided accurately within those tissues. But once they were placed into a dish, the frequency of chromosome mis-segregation drastically increased. The cells that were less tied to structures in their tissue were not affected by the lack of architecture in culture dishes.
The researchers found that maintaining the architectural conditions of the cell’s native environment is essential for chromosome stability. Cells removed from the context of their tissue don’t always faithfully represent natural processes.
The researchers determined that architecture didn’t have an obvious effect on the expression of known genes involved in segregation. The disruption in tissue architecture likely causes mechanical changes that disrupt segregation, in a manner that is independent of mutations or gene expression changes.
“It was surprising to us that for something so intrinsic to the cell — something that's happening entirely within the cell and so fundamental to the cell's existence — where that cell is sitting actually matters quite a bit,” Knouse says.
Through the Cancer Genome Project, scientists learned that despite high rates of chromosome mis-segregation, many cancers lack any mutations to the cellular machinery that controls chromosome partitioning. This left scientists searching for the cause of the increase of these division errors. This study suggests that tissue architecture could be the culprit.
Cancer development often involves disruption of tissue architecture, whether during tumor growth or metastasis. This disruption of the extracellular environment could trigger chromosome segregation errors in the cells within the tumor.
“I think [this paper] really could be the explanation for why certain kinds of cancers become chromosomally unstable,” says Iain Cheeseman, a professor of biology at MIT and a member of the Whitehead Institute, who was not involved in the study.
The results point not only to a new understanding of the cellular mechanical triggers and effects of cancers, but also to a new understanding of how cell biology must be studied.
“Clearly a two-dimensional culture system does not faithfully recapitulate even the most fundamental processes, like chromosome segregation,” Knouse says. “As cell biologists we really must start recognizing that context matters.”
This work was supported by the National Institutes of Health, the Kathy and Curt Marble Cancer Research Fund, and the Koch Institute Support (core) Grant from the National Cancer Institute.