A team of researchers at MIT and Children’s Hospital Boston has built cardiac patches studded with tiny gold wires that could be used to create pieces of tissue whose cells all beat in time, mimicking the dynamics of natural heart muscle. The development could someday help people who have suffered heart attacks.
The study, reported this week in Nature Nanotechnology, promises to improve on existing cardiac patches, which have difficulty achieving the level of conductivity necessary to ensure a smooth, continuous “beat” throughout a large piece of tissue.
“The heart is an electrically quite sophisticated piece of machinery,” says Daniel Kohane, a professor in the Harvard-MIT Division of Health Sciences and Technology (HST) and senior author of the paper. “It is important that the cells beat together, or the tissue won’t function properly.”
The unique new approach uses gold nanowires scattered among cardiac cells as they’re grown in vitro, a technique that “markedly enhances the performance of the cardiac patch,” Kohane says. The researchers believe the technology may eventually result in implantable patches to replace tissue that’s been damaged in a heart attack.
Co-first authors of the study are MIT postdoc Brian Timko and former MIT postdoc Tal Dvir, now at Tel Aviv University in Israel; other authors are their colleagues from HST, Children’s Hospital Boston and MIT’s Department of Chemical Engineering, including Robert Langer, the David H. Koch Institute Professor.
Ka-thump, ka-thump
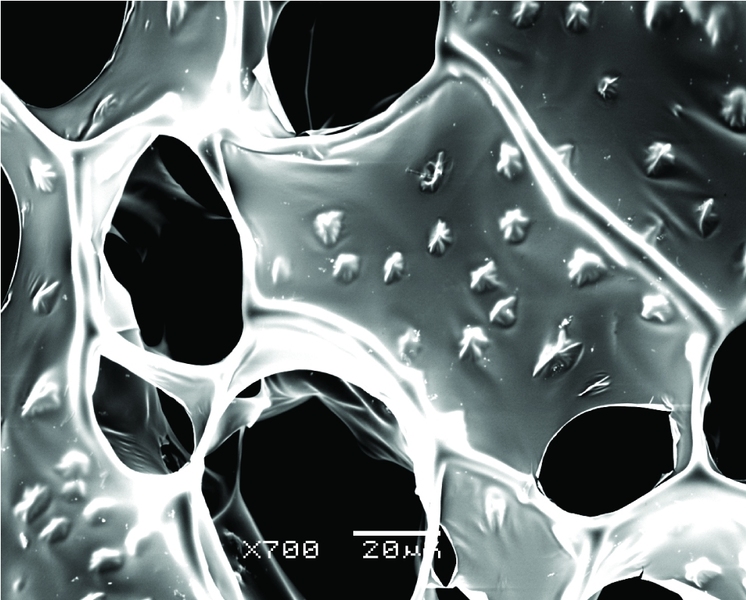
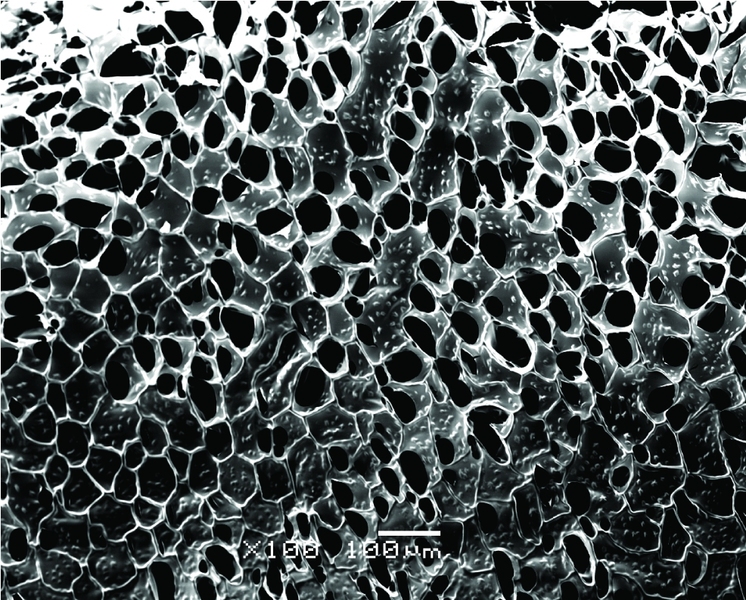
To build new tissue, biological engineers typically use miniature scaffolds resembling porous sponges to organize cells into functional shapes as they grow. Traditionally, however, these scaffolds have been made from materials with poor electrical conductivity — and for cardiac cells, which rely on electrical signals to coordinate their contraction, that’s a big problem.
“In the case of cardiac myocytes in particular, you need a good junction between the cells to get signal conduction,” Timko says. But the scaffold acts as an insulator, blocking signals from traveling much beyond a cell’s immediate neighbors, and making it nearly impossible to get all the cells in the tissue to beat together as a unit.
Video courtesy of the Disease Biophysics Group, Harvard University
To solve the problem, Timko and Dvir took advantage of their complementary backgrounds — Timko’s in semiconducting nanowires, Dvir’s in cardiac-tissue engineering — to design a brand-new scaffold material that would allow electrical signals to pass through.
“We started brainstorming, and it occurred to me that it’s actually fairly easy to grow gold nanoconductors, which of course are very conductive,” Timko says. “You can grow them to be a couple microns long, which is more than enough to pass through the walls of the scaffold.”
From micrometers to millimeters
The team took as their base material alginate, an organic gum-like substance that is often used for tissue scaffolds. They mixed the alginate with a solution containing gold nanowires to create a composite scaffold with billions of the tiny metal structures running through it.
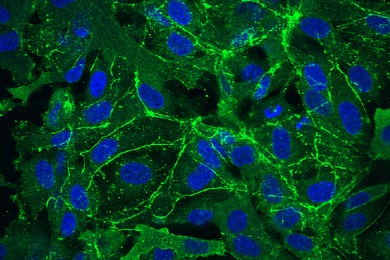
Then, they seeded cardiac cells onto the gold-alginate composite, testing the conductivity of tissue grown on the composite compared to tissue grown on pure alginate. Because signals are conducted by calcium ions in and among the cells, the researchers could check how far signals travel by observing the amount of calcium present in different areas of the tissue.
“Basically, calcium is how cardiac cells talk to each other, so we labeled the cells with a calcium indicator and put the scaffold under the microscope,” Timko says. There, they observed a dramatic improvement among cells grown on the composite scaffold: The range of signals conduction improved by about three orders of magnitude.
“In healthy, native heart tissue, you’re talking about conduction over centimeters,” Timko says. Previously, tissue grown on pure alginate showed conduction over only a few hundred micrometers, or thousandths of a millimeter. But the combination of alginate and gold nanowires achieved signal conduction over a scale of “many millimeters,” Timko says.
“It’s really night and day. The performance that the scaffolds have with these nanomaterials is just much, much better,” Kohane says.
“It’s very beautiful work,” says Charles Lieber, a professor of chemistry at Harvard University. “I think the results are quite unambiguous, and very exciting — both in showing fundamentally that they’ve improved the conductivity of these scaffolds, and then how that clearly makes a difference in enhancing the collective firing of the cardiac tissue.”
The researchers plan to pursue studies in vivo to determine how the composite-grown tissue functions when implanted into live hearts. Aside from implications for heart-attack patients, Kohane adds that the successful experiment “opens up a bunch of doors” for engineering other types of tissues; Lieber agrees.
“I think other people can take advantage of this idea for other systems: In other muscle cells, other vascular constructs, perhaps even in neural systems, this is a simple way to have a big impact on the collective communication of cells,” Lieber says. “A lot of people are going to be jumping on this.”
The study, reported this week in Nature Nanotechnology, promises to improve on existing cardiac patches, which have difficulty achieving the level of conductivity necessary to ensure a smooth, continuous “beat” throughout a large piece of tissue.
“The heart is an electrically quite sophisticated piece of machinery,” says Daniel Kohane, a professor in the Harvard-MIT Division of Health Sciences and Technology (HST) and senior author of the paper. “It is important that the cells beat together, or the tissue won’t function properly.”
The unique new approach uses gold nanowires scattered among cardiac cells as they’re grown in vitro, a technique that “markedly enhances the performance of the cardiac patch,” Kohane says. The researchers believe the technology may eventually result in implantable patches to replace tissue that’s been damaged in a heart attack.
Co-first authors of the study are MIT postdoc Brian Timko and former MIT postdoc Tal Dvir, now at Tel Aviv University in Israel; other authors are their colleagues from HST, Children’s Hospital Boston and MIT’s Department of Chemical Engineering, including Robert Langer, the David H. Koch Institute Professor.
Ka-thump, ka-thump
To build new tissue, biological engineers typically use miniature scaffolds resembling porous sponges to organize cells into functional shapes as they grow. Traditionally, however, these scaffolds have been made from materials with poor electrical conductivity — and for cardiac cells, which rely on electrical signals to coordinate their contraction, that’s a big problem.
“In the case of cardiac myocytes in particular, you need a good junction between the cells to get signal conduction,” Timko says. But the scaffold acts as an insulator, blocking signals from traveling much beyond a cell’s immediate neighbors, and making it nearly impossible to get all the cells in the tissue to beat together as a unit.
Video courtesy of the Disease Biophysics Group, Harvard University
To solve the problem, Timko and Dvir took advantage of their complementary backgrounds — Timko’s in semiconducting nanowires, Dvir’s in cardiac-tissue engineering — to design a brand-new scaffold material that would allow electrical signals to pass through.
“We started brainstorming, and it occurred to me that it’s actually fairly easy to grow gold nanoconductors, which of course are very conductive,” Timko says. “You can grow them to be a couple microns long, which is more than enough to pass through the walls of the scaffold.”
From micrometers to millimeters
The team took as their base material alginate, an organic gum-like substance that is often used for tissue scaffolds. They mixed the alginate with a solution containing gold nanowires to create a composite scaffold with billions of the tiny metal structures running through it.
Then, they seeded cardiac cells onto the gold-alginate composite, testing the conductivity of tissue grown on the composite compared to tissue grown on pure alginate. Because signals are conducted by calcium ions in and among the cells, the researchers could check how far signals travel by observing the amount of calcium present in different areas of the tissue.
“Basically, calcium is how cardiac cells talk to each other, so we labeled the cells with a calcium indicator and put the scaffold under the microscope,” Timko says. There, they observed a dramatic improvement among cells grown on the composite scaffold: The range of signals conduction improved by about three orders of magnitude.
“In healthy, native heart tissue, you’re talking about conduction over centimeters,” Timko says. Previously, tissue grown on pure alginate showed conduction over only a few hundred micrometers, or thousandths of a millimeter. But the combination of alginate and gold nanowires achieved signal conduction over a scale of “many millimeters,” Timko says.
“It’s really night and day. The performance that the scaffolds have with these nanomaterials is just much, much better,” Kohane says.
“It’s very beautiful work,” says Charles Lieber, a professor of chemistry at Harvard University. “I think the results are quite unambiguous, and very exciting — both in showing fundamentally that they’ve improved the conductivity of these scaffolds, and then how that clearly makes a difference in enhancing the collective firing of the cardiac tissue.”
The researchers plan to pursue studies in vivo to determine how the composite-grown tissue functions when implanted into live hearts. Aside from implications for heart-attack patients, Kohane adds that the successful experiment “opens up a bunch of doors” for engineering other types of tissues; Lieber agrees.
“I think other people can take advantage of this idea for other systems: In other muscle cells, other vascular constructs, perhaps even in neural systems, this is a simple way to have a big impact on the collective communication of cells,” Lieber says. “A lot of people are going to be jumping on this.”