A surprising MIT study published in Nature at the end of 2016 helped to spur interest in the possibility that light flickering at the frequency of a particular gamma-band brain rhythm could produce meaningful therapeutic effects for people with Alzheimer’s disease. In a new review paper in the Journal of Internal Medicine, the lab that led those studies takes stock of what a growing number of scientists worldwide have been finding out since then in dozens of clinical and lab benchtop studies.
Brain rhythms (also called brain “waves” or “oscillations”) arise from the synchronized network activity of brain cells and circuits as they coordinate to enable brain functions such as perception or cognition. Lower-range gamma-frequency rhythms, those around 40 cycles a second, or hertz (Hz), are particularly important for memory processes, and MIT’s research has shown that they are also associated with specific changes at the cellular and molecular level. The 2016 study and many others since then have produced evidence, initially in animals and more recently in humans, that various noninvasive means of enhancing the power and synchrony of 40Hz gamma rhythms helps to reduce Alzheimer’s pathology and its consequences.
“What started in 2016 with optogenetic and visual stimulation in mice has expanded to a multitude of stimulation paradigms, a wide range of human clinical studies with promising results, and is narrowing in on the mechanisms underlying this phenomenon,” write the authors including Li-Huei Tsai, Picower Professor in The Picower Institute for Learning and Memory and the Department of Brain and Cognitive Sciences at MIT.
Though the number of studies and methods has increased and the data have typically suggested beneficial clinical effects, the article’s authors also clearly caution that the clinical evidence remains preliminary and that animal studies intended to discern how the approach works have been instructive, but not definitive.
“Research into the clinical potential of these interventions is still in its nascent stages,” the researchers, led by MIT postdoc Cristina Blanco-Duque, write in introducing the review. “The precise mechanisms underpinning the beneficial effects of gamma stimulation in Alzheimer’s disease are not yet fully elucidated, but preclinical studies have provided relevant insights.”
Preliminarily promising
The authors list and summarize results from 16 clinical studies published over the last several years. These employ gamma-frequency sensory stimulation (e.g., exposure to light, sound, tactile vibration, or a combination); transcranial alternating current stimulation (tACS), in which a brain region is stimulated via scalp electrodes; or transcranial magnetic stimulation (TMS), in which electric currents are induced in a brain region using magnetic fields. The studies also vary in their sample size, design, duration, and in what effects they assessed. Some of the sensory studies using light have tested different colors and different exact frequencies. And while some studies show that sensory stimulation appears to affect multiple regions in the brain, tACS and TMS are more regionally focused (though those brain regions still connect and interact with others).
Given the variances, the clinical studies taken together offer a blend of uneven but encouraging evidence, the authors write. Across clinical studies involving patients with Alzheimer’s disease, sensory stimulation has proven safe and well-tolerated. Multiple sensory studies have measured increases in gamma power and brain network connectivity. Sensory studies have also reported improvements in memory and/or cognition, as well as sleep. Some have yielded apparent physiological benefits such as reduction of brain atrophy, in one case, and changes in immune system activity in another. So far, sensory studies have not shown reductions in Alzheimer’s hallmark proteins, amyloid or tau.
Clinical studies stimulating 40Hz rhythms using tACS, ranging in sample size from only one to as many as 60, are the most numerous so far, and many have shown similar benefits. Most report benefits to cognition, executive function, and/or memory (depending sometimes on the brain region stimulated), and some have assessed that benefits endure even after treatment concludes. Some have shown effects on measures of tau and amyloid, blood flow, neuromodulatory chemical activity, or immune activity. Finally, a 40Hz stimulation clinical study using TMS in 37 patients found improvements in cognition, prevention of brain atrophy, and increased brain connectivity.
“The most important test for gamma stimulation is without a doubt whether it is safe and beneficial for patients,” the authors write. “So far, results from several small trials on sensory gamma stimulation suggest that it is safe, evokes rhythmic EEG brain responses, and there are promising signs for AD [Alzheimer's disease] symptoms and pathology. Similarly, studies on transcranial stimulation report the potential to benefit memory and global cognitive function even beyond the end of treatment.”
Studying underlying mechanisms
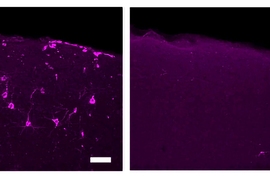
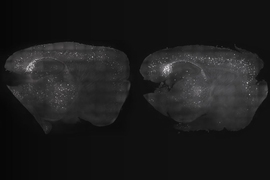
In parallel, dozens more studies have shown significant benefits in mice including reductions in amyloid and tau, preservation of brain tissue, and improvements in memory. But animal studies also have offered researchers a window into the cellular and molecular mechanisms by which gamma stimulation might have these effects.
Before MIT’s original studies in 2016 and 2019, researchers had not attributed molecular changes in brain cells to changes in brain rhythms, but those and other studies have now shown that they affect not only the molecular state of neurons, but also the brain’s microglia immune cells, astrocyte cells that play key roles in regulating circulation, and indeed the brain’s vasculature system. A hypothesis of Tsai’s lab right now is that sensory gamma stimulation might promote the clearance of amyloid and tau via increased circulatory activity of brain fluids.
A hotly debated aspect of gamma stimulation is how it affects the electrical activity of neurons, and how pervasively. Studies indicate that inhibitory “interneurons” are especially affected, though, offering a clue about how increased gamma activity, and its physiological effects, might propagate.
“The field has generated tantalizing leads on how gamma stimulation may translate into beneficial effects on the cellular and molecular level,” the authors write.
Gamma going forward
As the authors make clear that more definitive clinical studies are needed, they note that at the moment, there are now 15 new clinical studies of gamma stimulation underway. Among these is a phase 3 clinical trial by the company Cognito Therapeutics, which has licensed MIT’s technology. That study plans to enroll hundreds of participants.
Meanwhile, some recent or new clinical and preclinical studies have begun looking at whether gamma stimulation may be applicable to neurological disorders other than Alzheimer’s, including stroke or Down syndrome. In experiments with mouse models, for example, an MIT team has been testing gamma stimulation’s potential to help with cognitive effects of chemotherapy, or “chemobrain.”
“Larger clinical studies are required to ascertain the long-term benefits of gamma stimulation,” the authors conclude. “In animal models the focus should be on delineating the mechanism of gamma stimulation and providing further proof of principle studies on what other applications gamma stimulation may have.”
In addition to Tsai and Blanco-Duque, the paper’s other authors are Diane Chan, Martin Kahn, and Mitch Murdock.