Interdisciplinary materials research holds the key to solving the existential challenges facing humanity, former Sandia National Laboratories executive Julia M. Phillips told researchers who gathered for the annual MIT Materials Research Laboratory (MRL) Materials Day Symposium last month.
“What is both very exciting for us as materials researchers, also a little frustrating, is that the real impact of materials occurs when they turn into something that you actually carry around in your pocket or whatever,” Phillips said at the Oct. 11 event.
During the second half of the 20th century, many of the technological advances that we take for granted today, such as laptop computers and smart phones, came from fundamental advances in materials research and the ability to control and make materials, she said. Phillips, who retired from Sandia National Laboratories as vice president and chief technology officer, also serves as chair of the MRL External Advisory Board and is a member of the National Science Board.
MRL was formed from the merger of the Materials Processing Center and the Center for Materials Science and Engineering, which became effective on Oct. 1. In his introductory remarks, MRL Director Carl V. Thompson noted the appointment of Geoffrey S.D. Beach, an associate professor of materials science and engineering as co-director of the MRL and principal investigator for the National Science Foundation Materials Research Science and Engineering Center.
Fueled by industrial needs and government-funded research in the post-World War II era, “materials research was undeniably an early model for interdisciplinary research,” Phillips said. With new tools such as scanning probe microscopes to understand the structure and properties of materials, materials scientists in the last half of the 20th century created whole new classes of materials and products, ranging from super alloys that enabled larger and more reliable jet engines to strained layer superlattices that underlie modern magnetic recording, lasers, and infrared detectors.
Future gains will come from the ability to synthesize and control increasingly complex materials, Phillips said, noting progress in areas such as high-temperature superconductors, porous solids like metal organic frameworks, and metamaterials that generate new properties from combining biological materials, organics, ceramic, and metals at near molecular scale precision in ways not found in nature.
“Somewhere in the fuzzy space between molecules and materials,” these newer materials have very interesting properties that are still in the process of being fully explored, and they will be exploited in the years to come, Phillips noted. “It’s very clear to many people that these also will be transformational as we move forward.”
The materials research approach, which brings together researchers from across different science and engineering fields to solve complex problems, provides a model for solving 21st century challenges in energy, environment and sustainability; health care and medicine; vulnerability to human and natural threats; and expanding and enhancing human capability and joy. “These are exemplars, but you can see materials written all over this list, and I would posit that any comparable list you might come up with would have materials written all over it,” Phillips said. “In order to address those grand challenges, we really need to be able to treat realistically complex systems that bring together all of these disciplines from the sciences, from engineering, from the social and behavioral sciences, and arguably even from the arts.”
Progress in scientific understanding and computational modeling are accelerating researchers’ ability to predict the structure and properties of new materials before actually making them, Phillips said.
MIT faculty members Antoine Allanore, Polina Anikeeva, A. John Hart, Pablo Jarillo-Herrero, Juejun Hu, and Jennifer Rupp presented research updates on their recent work which spans a range from ultra-thin layered materials for new electronic devices and cellular level probes for the brain and spinal cord to larger scale methods for 3D printing and metals processing.
Merging 2-D materials with CMOS
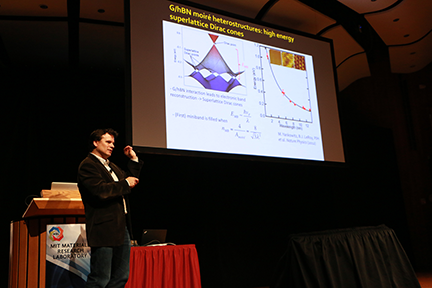
Associate professor of physics Pablo Jarillo-Herrero stacks atomically thin, two-dimensional layers of different materials to discover new properties. Jarillo-Herrero’s lab demonstrated photodetectors, solar cells and the world’s thinnest LED. With materials such as tungsten selenide, changing the number of layers also changes their electronic properties. Although graphene itself has no bandgap, closely aligning the lattices of graphene and boron nitride opens a 30-millivolt bandgap in graphene, he said.
“You have full electronic control with gate voltages,” Jarillo-Herrero said. Using bilayer molybdenum ditelluride, which is 10,000 times thinner than a silicon solar cell, he showed in work published in Nature Nanotechnology, a photodetector just 10 nanometers thick can be integrated on a silicon photonic crystal waveguide.
“You can just stack this at the very end of your CMOS [complementary metal oxide semiconductor] processing, and you don’t have to do any extra fabrication, any extra growth, you can just slap it on top,” Jarillo-Herrero explained. “It can be made as thin as 4 nanometers, so it’s still ultra thin, and you have a high degree of control in an ultra thin platform. The whole thing is semitransparent so we can see the light go in and out.” These new devices can be operated at telecommunications wavelengths by tuning the bandgap of the material.
Phase change materials
Juejun "JJ" Hu, the Merton C. Flemings Associate Professor of Materials Science and Engineering, is reducing power consumption, shrinking device size, and ramping up processing speed with innovative combinations of materials that alternate between two different solid states, or phases, such as an alloy of germanium, antimony, selenium, and tellurium. These materials are the basis for nonvolatile storage, meaning their memory state is preserved even when the power is turned off. Hu collaborated with Professor Jeffrey C. Grossman and former postdoc Huashan Li to identify desirable materials for these alloys from first principles calculations, and graduate materials science and engineering student Yifei Zhang did much of the experimental work.
An earlier generation of devices based on germanium, antimony, and tellurium (GST) suffers from losses to light absorption by the material. To overcome this problem, Hu substituted some of the tellurium with a lighter element, selenium, creating a new four-element structure of germanium, antimony, selenium and tellurium (GSST). “We increase the bandgap to suppress short wavelength absorption, and we actually minimize any carrier mobility to mitigate the free carrier absorption,” he explained. Switching between amorphous and crystalline states can be triggered with a laser pulse or an electrical signal.
Although the structural state switching happens on the order of 100 nanoseconds, figuring out the techniques to accomplish it took a year of work, Hu said. Specifically, he found that using materials that switch between amorphous and crystalline states allows light to be directed over two different paths and reduces power consumption. He coupled this GSST optical phase change material with silicon nitride microresonators and waveguides to show this behavior. These switches based on phase change materials can be connected in a matrix to enable variable light control on a chip. Ultimately, Hu hopes to use this technology to build re-programmable photonic integrated circuits.
New tools for brain exploration
Class of 1942 Associate Professor in Materials Science and Engineering Polina Anikeeva works at the border between synthetic devices and the nervous system. Traditional electronic devices, with hardness like a knife, can trigger a foreign-body response from brain tissue, which typically is as soft as pudding or yogurt. Working with Prof. Yoel Fink and other MIT colleagues, Anikeeva developed soft polymer-based devices to stimulate and record activity of brain and spinal cord tissue borrowing from optical fiber drawing techniques.
An early version of their multi-functional fibers included three key elements: conductive polyethylene carbon composite electrodes to record brain cell activity; a transparent polycarbonate waveguide with cyclic olefin copolymer cladding to deliver light; and microfluidic channels to deliver drugs.
“Using this structure, for the first time, we were able to record, stimulate and pharmacologically modulate neural activity,” Anikeeva said. But the device recorded activity from clusters of neurons, not individual neurons. Anikeeva and her team addressed this problem by integrating graphite into the polyethylene composite electrodes, which increased their conductivity enough to shrink them into a structure that is as thin as a human hair. The device has six electrodes, an optical waveguide and two microfluidic channels.
Yet adding graphite increased the size and hardness of the glassy polycarbonate device, so her group turned to a new process using rubbery, stretchy polymers that they then coated with a conductive metal nanowire mesh. “This mesh of conductive metal nanowires can maintain low impedance even at 100 percent strain, and it maintains its structural integrity without any changes up to 20 percent strain, which is sufficient for us to operate in the spinal cord,” Anikeeva said.
Her students implanted these nanowire-mesh coated fibers in mice, which allowed them to stimulate and record neural activity in the spinal cord. A video showed a mouse moving its hindlimb when an optical signal delivered to the lumbar spinal cord traveled down the sciatic nerve to the gastrocnemius muscle. In these experiments, the device implanted in mice showed no decline in performance a year after surgery, Anikeeva said.
More recently, Anikeeva developed iron oxide-based nanoparticles that heat up in an applied magnetic field, which can trigger a response from neurons in the brain that express ion channels that are sensitive to heat such as capsaicin receptor, the same mechanism that is triggered when we eat hot peppers. Experimenting with mice, Anikeeva injected these tiny particles deep in the brain in a section that is associated with reward.
“In our lab, we have started by modeling hysteresis in magnetic nanoparticles, synthesizing a broad range of these nanomaterials by engineering iron oxide with dopants and looking at different sizes and shapes, developing power electronics and a biological tool kit to assess this process,” Anikeeva explained. “In this case, there is no external hardwire, no wires, no implants, nothing is sticking out of the brain. … However, they can now perceive magnetic field.” she said. To quantify their results, the researchers measured calcium ion influx into neurons. Work is now focused on shortening the response time to a few thousandths of a second by improving the heat output of the magnetic nanoparticles.
Ceramics for solid-state batteries, carbon dioxide sensors, and memristive computing
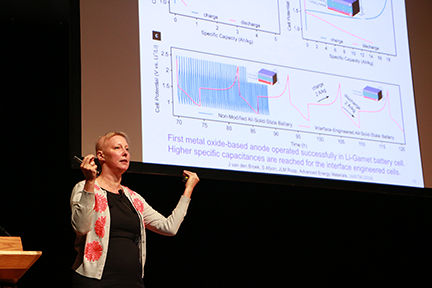
Jennifer L. M. Rupp, the Thomas Lord Assistant Professor of Materials Science and Engineering, presented research showing a solid lithium garnet electrolyte can lead to batteries miniaturized on an integrated circuit chip.
Safety concerns regarding lithium batteries stem from their liquid component, which serves as the electrolyte and presents a risk of catching fire in air. Replacing the liquid electrolyte with a solid one could make batteries safer, Rupp explained. Her research shows that a ceramic material made of garnet, a material that is perhaps more familiar as a gemstone, can effectively pass lithium through a battery cell, but because it is solid, can be very safe for batteries and also have the opportunity to be miniaturized to thin film architectures. This garnet is a four-element compound of lithium, lanthanum, zirconium and oxygen.
“The lithium is completely encapsulated; there is no risk of inflammation,” Rupp said.
In published research, Rupp showed that pairing a lithium titanium oxide anode with a ceramic garnet electrolyte and blurring the interface between the two materials allowed much faster battery charging time for large-scale cells. Lessons learned from applying these garnet materials pointed also to a new use for carbon dioxide sensing.
“We can reconfigure the electrodes to have one electrode which simply goes as a reference, and another which undergoes a chemical reaction with carbon dioxide, and we use a tracker potential to track the effective change of carbon dioxide concentration in the environment based on bulk processing,” she explained. Rupp is also developing strained multi-layer materials to improve storage for memristive memory and computing elements.
Frontier for metals at high temperature
Associate Professor of Metallurgy Antoine Allanore pointed out that from 1980 to 2010, the world almost doubled its consumption of materials, with the fastest growth in metals and minerals. Such demand is due to the formidable low cost and high productivity of materials processing. The majority of such processes involve at some stage a high temperature operation and often the molten state of matter. Developing the science and engineering of the molten state brings huge opportunities, for example heat management in high-temperature processes such as metals extraction and glass making.
Steelmaking, for example, is already a highly efficient manufacturing process, turning out rebar, coil or wires of steel at a cost less than 32 cents per kilogram. “Productivity is actually the key criteria to make materials processing successful and matter at the scale of the challenge of adding 2 billion people in the next 20 years,” he said.
Allanore’s group demonstrated that tin sulfide at high temperature, about 1,130 degrees Celsius, is an effective thermoelectric generator. “We have indications that the theoretical figure of merit for some sulfides, can be up to 1 at 1,130 [degrees Celsius]. For molten copper sulfide for example, we have estimates of the thermal conductivity, the melting point, and we have a cost that is a little bit high in my opinion, but that’s the nature of the research,” Allanore said. When his group looked at existing data, they found that for many molten compounds of sulfur and a metal, such as tin, lead or nickel, the thermoelectric figure of merit, as well as the compositional phases, had never been quantified, opening a frontier for new materials science research at high temperature.
“It’s actually very difficult to know what are the true properties of the liquid,” Allanore said. “I need to know if that material will have semiconductivity. I need to know if it’s going to be denser or lighter than another liquid. … We don’t actually have computational methods to predict such property for liquids at high temperature.”
To address the problem, Allanore studied the relation in high-temperature melts between transport properties, including electrical conductivity and Seebeck coefficients, and a thermodynamic property called entropy. “We’ve put together a theoretical model that connects the transport property, like thermal power, and the thermodynamic property like entropy. This is important because it works for semiconductors, it works for metallic materials and more importantly it allows to find out regions of immiscibility in liquids,” Allanore said. Immiscibility means a material in the given condition will separate into two phases that do not mix together and remain separate.
Allanore has also developed a new method for observing molten compounds such as alumina, using a floating zone furnace, which is a transparent quartz tube located at the focal distance of four lamps. “If we can do that with oxides, we would really like to do that with sulfides,” he explained, showing a picture of molten tin sulfide sitting on a graphite plate in the floating zone furnace. The wide range of temperatures and properties of molten materials, “the ultimate state of condensed matter”, allows for better heat management, higher processing temperatures and electricity harvesting or electrical control of heat flow, he said.
3-D printing a new manufacturing model
Traditional manufacturing requires economies of scale, in particular, large production volumes because of the fixed costs necessary to set up the production process, but 3-D printing and other additive manufacturing technologies offer an alternative of high-performance, customizable products and devices, Associate Professor of Mechanical Engineering A. John Hart said.
Additive manufacturing is already a $6 billion a year business with reach from Hollywood special effects to high-tech jet engine nozzles. “Additive manufacturing already enables a diverse collection of materials, applications, and related processes – including by extrusion of plastics, melting metals, using lasers, and by coordinated chemical reactions that essentially are done with point wise control,” Hart explained.
“We can think of accessing new spaces in terms of the value of the products we create using additive manufacturing, also generally known as 3-D printing. 3-D printing is reshaping the axes by which we judge the economic viability of a manufacturing process, and allowing us to access new value spaces. For instance, we can think not only about production volume, but think about advantages in complexity of geometries, and advantages by customization of products to specific markets or even individuals. In these ways, 3-D printing is influencing the entire product life cycle,” Hart said.
For instance, Hart’s group studied existing 3-D printers to discover how to speed up the process from about 60 minutes to just 5 to 10 minutes to print a handheld mechanical part such as a gear. Former graduate student Jamison Go, SM '15 led this work, Hart said, building a desktop 3-D printer about the size of a small microwave oven. The system features a control system for the printhead that moves the motors to the corner; an extrusion mechanism that drives the feedstock polymer filament like a screw; and a laser that penetrates and melts the polymer.
“By combining the fast motion control, the high heat transfer, and the high force, we can overcome the limits of the existing system,” Hart explained. The new design is three to 10 times faster in build rate than existing machines.
“These kinds of steps forward can also change how we think about producing objects. If you can make something fast, you can think about how you might, or how others might, work differently,” he said. He mentioned, for instance, physicians who may need to 3-D print a part for an emergency medical operation, or a repair technician who could use a 3-D printer rather than hold inventory of many spare parts.
Hart’s group is currently working in collaboration with Oak Ridge National Lab on algorithms for optimization of 3-D printing toolpaths, and adapting his innovations to large-scale 3-D printers. “We can think about upscaling these principles to high productivity systems that are not only printing small things but printing big things,” Hart said.
Hart has also worked with 3-D printing of cellulose, which can be used for customization of consumer products and antimicrobial devices, and is the world’s most abundant natural polymer. He co-founded the company Desktop Metal with three other MIT faculty members and Ric Fulop, SL ’06, who serves as Desktop Metal’s CEO. “The company is only two years old and will soon ship its first product which enables an entirely new approach to metal 3-D printing,” Hart said.