Under a microscope, a cell’s cytoplasm can resemble a tiny underwater version of New York’s Times Square: Thousands of proteins swarm through a cytoplasm’s watery environment, coming together and breaking apart like a cytoskeletal flash mob.
Organelles such as mitochondria and lysosomes must traverse this crowded, ever-changing cytoplasmic space to deliver materials to various parts of a cell.
Now engineers at MIT have found that these organelles and other intracellular components may experience the surrounding cytoplasm as very different environments as they travel. For instance, a cell’s nucleus may “feel” the cytoplasm as a fluid, honey-like material, while mitochondria may experience it more like toothpaste.
The team, led by Ming Guo, the Brit and Alex d'Arbeloff Career Development Assistant Professor in MIT’s Department of Mechanical Engineering, found that an organelle feels a certain resistance in cytoplasm, depending on that organelle’s size and the speed at which it moves through a cell. In particular, these characteristics determine how easily it can push against a cytoplasm’s surrounding water and move through its ever-changing web of cytoskeletal protein structures.
Certain organelles may have to work harder to make their way through cytoplasm, and may therefore feel more resistance. The researchers found that the resistance that any major organelle may feel ranges from that of a viscous fluid to an elastic, rubbery solid.
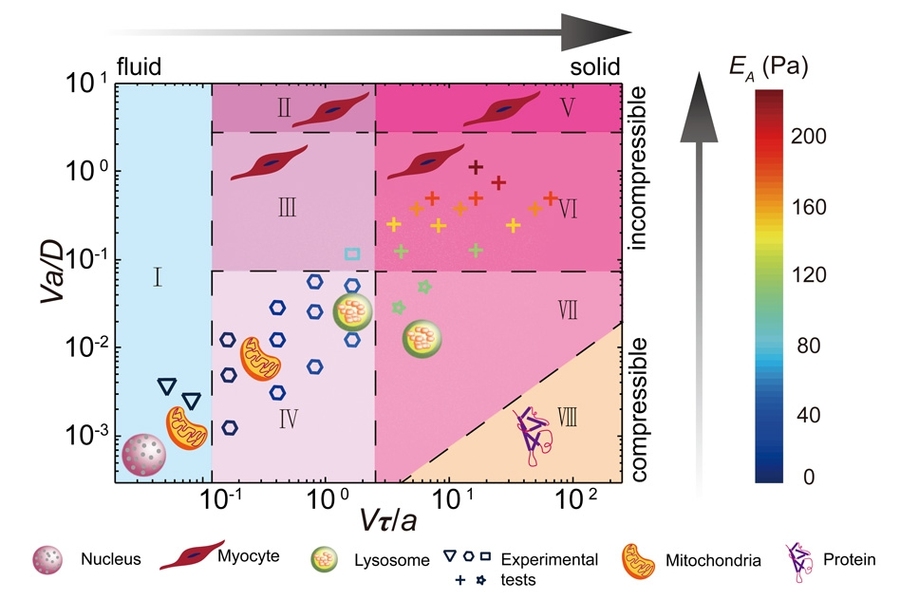
Guo and his colleagues have drawn up a phase diagram to describe the type of material that a cytoplasm would resemble, from the perspective of an organelle, given the organelle’s size and speed.
“Our main goal was to provide the most fundamental understanding of living cells as a material,” Guo says. “With this phase diagram, as long as you tell me the size and speed at which an organelle moves, I can tell you what mechanical environment it sees.”
The results, published this week in the Proceedings of the National Academy of Sciences, may help guide pharmaceutical designs. For instance, with the team’s phase diagram, scientists can tailor a drug’s size to enable it to travel within a cell with a certain amount of ease.
“A drug with a 100-nanometer diameter will feel a very different resistance than something that is 500 nanometers wide,” Guo says. “This can be a guide to understanding how a drug is delivered and transported inside a cell.”
The study’s lead author is Jiliang Hu, a former visiting student at MIT, who will join Guo’s lab as a graduate student this fall. Other co-authors include Yulong Han, a postdoc in Guo’s lab; and Alan Grodzinsky, professor of biological engineering, electrical engineering and computer science, and mechanical engineering at MIT; along with Somaye Jafari and Shengqiang Cai at the University of California at San Diego.
What a drag
Most scientists who study the transport of materials within a cell have focused on the drivers of that transport — namely, molecular motors, a family of biological agents that actively convert a cell’s energy into mechanical work to move cargo across a cell.
“But as mechanical engineers, we think the driving force is not the only part of this transport process, but that resistance of the surrounding material is actually equally important,” Guo says. “For example, it’s not just your own energy that determines how you move through a crowd — the mechanical resistance of the crowd itself can also affect your movement.”
In the case of living cells, Guo wondered whether the surrounding cytoplasm would have a similar crowding effect on the movement of major organelles such as mitochondria and lysosomes.
To test his hypothesis, he and his colleagues carried out experiments on living mammalian cells, into which they injected tiny plastic beads ranging in size from 0.5 to 1.5 microns — a range that covers most major organelles. They then dragged each bead across a cell using optical tweezers, a technique that employs a highly focused laser beam to physically move microscopic objects.
The researchers trapped and pulled each bead toward the cell edge at a constant speed and measured the force required to drag the bead a certain distance. They interpreted that force as the mechanical resistance of the surrounding cytoplasm.
They then assumed that a cytoplasm’s mechanical resistance stems from two main sources: poroelasticity and viscoelasticity. Poroelasticity originates from how fast cytoplasm can diffuse water out of a region. The group reasoned that the more poroelastic cytoplasm is, the more effort an object such as an organelle needs to make to push water out of its way.
Viscoelasticity, in the context of cytoplasm, is how fast its cytoskeleton, or web of proteins, changes configuration. A cell’s cytoskeleton serves as a sort of scaffold, made from thousands of proteins that are constantly assembling, disassembling, and reassembling. This dynamic network can feel like both an elastic solid and a viscous fluid. The faster a cytoskeleton rearranges itself, the more fluid-like it is. The researchers reasoned that an organelle would feel less resistance while moving through a more fluid-like, frequently changing cytoskeleton.
It’s all about perspective
Guo and his colleagues analyzed their experimental results and found that a bead’s size and speed were related to the type of resistance that it encountered as it was dragged across a cell. In general, the larger the beads, the more they met with poroelastic resistance, as large beads with greater surface area have to push against more water to move themselves through.
On the other hand, the faster a bead was dragged, the more it met with a solid-like resistance. As Guo explains it, “the faster you move, the more permanent [cytoskeletal] structures you would see and feel resistance to.”
The researchers drew up their phase diagram based on their experimental results. They then looked through the scientific literature for speed and size measurements, made by others, of actual organelles in living cells. They plotted these measurements onto the diagram and found that, given their size and speed, these organelles should experience a range of resistances within cytoplasm.
“If you ask a nucleus, they would tell you the cytoplasm is like honey, because they are really large and slow, and they don’t feel cytoskeletal structures — they only feel the viscous disassembled protein solution, and have very small resistance,” Guo says. “But mitochondria would say it’s like toothpaste, because they are smaller and faster, and are sometimes blocked by these constantly changing structures. A lysosome, which is even smaller and faster, would tell you the cytoplasm is actually Jell-O, because they are moving so fast, they are constantly bouncing off these structures and meeting with resistance, like rubber. So their views are limited by their own size and speed.”
Guo hopes scientists will use the group’s phase diagram to characterize other cellular components, to understand how they see their cytoplasmic surroundings.
“People can use other parameters to find out what section of the phase diagram different organelles should belong to,” Guo says. “This will tell you what kind of distinct material they would feel.”