During about 30 percent of all heart attacks, the patient experiences no symptoms. However, unmistakable signs of the attack remain in the bloodstream for days. MIT researchers, working with Massachusetts General Hospital’s Cardiovascular Research Center, have now designed a tiny implant that can detect those signs, which could help doctors more rapidly determine whether a patient has had a heart attack.
In a study of mice, the team showed that the new implants can detect three proteins whose levels spike after a heart attack. Such devices could be used to monitor patients who are at high risk of heart attack, allowing doctors to respond more quickly if an attack occurs, preventing more severe heart disease from developing.
Most surprisingly, the researchers discovered that the sensors not only detect the proteins, they also reveal how much protein has ever been present. This is useful because it allows biomarkers (biological molecules that indicate a disease state) to be detected even if they are no longer in the bloodstream, says Michael Cima, professor of materials science and engineering and senior author of a paper on the work appearing in the Feb. 13 issue of Nature Biotechnology.
The new paper builds on earlier work. In 2009, Cima and others reported on a sensor able to detect human chorionic gonadotropin, a hormone overproduced in some cancer cells. The new study demonstrates that the sensor technology can work with a range of other molecules at concentrations found in the body.
Cima and his colleagues decided to modify their sensor to work with a disease that has very well defined biomarkers. They teamed up with MGH cardiologist Dr. Paul Huang, professor at Harvard Medical School, to look at heart disease because it has already been established that three proteins — myoglobin, cardiac troponin I and creatine kinase — peak in a characteristic pattern after a heart attack.
“If you go to the ER thinking you’ve had a heart attack, they take a blood sample and analyze it for these specific proteins,” says Cima. “If you think about cancer, there aren’t generally agreed-upon markers.”
The three co-first authors on the Nature Biotechnology paper are all graduate students: Yibo Ling (Cima lab), Terrence Pong (Huang lab) and Christophoros Vassiliou (Cima lab).
Specific detection
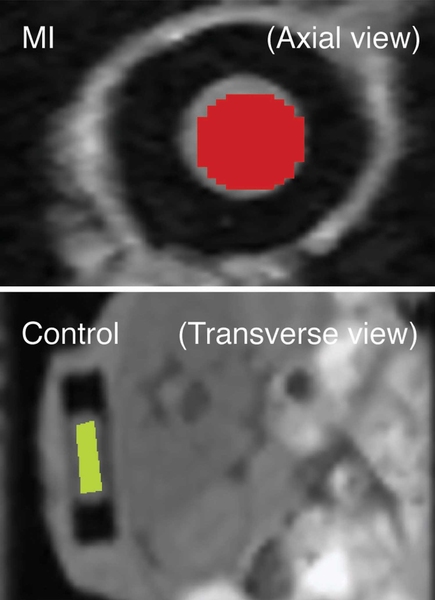
The small disk-shaped implant, which is 2 millimeters thick and 8 millimeters wide, contains iron-oxide particles coated with antibodies that target a specific biomarker. A semi-permeable membrane allows the protein targets to enter the device, where they bind to the antibodies. In this study, the researchers implanted six sensors — two for each heart-attack biomarker — under the skin of each mouse, and read them using magnetic resonance imaging (MRI).
One important finding is that all three sensors’ output was shown to be proportional to the size of the damage to the heart, says Huang. Thus, not only can they potentially be used to detect a heart attack, but they could yield some quick information on its severity.
This study marks the first time anyone has used implantable sensors to detect three different biomarkers, says Lee Josephson, associate professor at MGH’s Center for Molecular Imaging Research. “This shows how generalizable this technique is,” says Josephson, who was not involved in this study. Potential applications include not only detecting heart disease and cancer, but also tracking glucose levels in diabetic patients, he says.
Cima is now developing an implant that measures pH (acidity level), which could be useful for detecting heart disease or cancer. (Tumors are more acidic than healthy tissue, and dramatic increase in acidity is a near-instantaneous indicator of heart attack.)
In the future, he hopes to modify these sensors to detect low levels of hard-to-detect bacteria or viruses, or migrating tumor cells. “This may be a way to look for extremely small, or extremely transient concentrations of biological markers,” he says.
The current version of the implant is usable for about two months, but Cima believes the devices could be made to last longer by using antibodies that don’t break down as quickly.
In a study of mice, the team showed that the new implants can detect three proteins whose levels spike after a heart attack. Such devices could be used to monitor patients who are at high risk of heart attack, allowing doctors to respond more quickly if an attack occurs, preventing more severe heart disease from developing.
Most surprisingly, the researchers discovered that the sensors not only detect the proteins, they also reveal how much protein has ever been present. This is useful because it allows biomarkers (biological molecules that indicate a disease state) to be detected even if they are no longer in the bloodstream, says Michael Cima, professor of materials science and engineering and senior author of a paper on the work appearing in the Feb. 13 issue of Nature Biotechnology.
The new paper builds on earlier work. In 2009, Cima and others reported on a sensor able to detect human chorionic gonadotropin, a hormone overproduced in some cancer cells. The new study demonstrates that the sensor technology can work with a range of other molecules at concentrations found in the body.
Cima and his colleagues decided to modify their sensor to work with a disease that has very well defined biomarkers. They teamed up with MGH cardiologist Dr. Paul Huang, professor at Harvard Medical School, to look at heart disease because it has already been established that three proteins — myoglobin, cardiac troponin I and creatine kinase — peak in a characteristic pattern after a heart attack.
“If you go to the ER thinking you’ve had a heart attack, they take a blood sample and analyze it for these specific proteins,” says Cima. “If you think about cancer, there aren’t generally agreed-upon markers.”
The three co-first authors on the Nature Biotechnology paper are all graduate students: Yibo Ling (Cima lab), Terrence Pong (Huang lab) and Christophoros Vassiliou (Cima lab).
Specific detection
The small disk-shaped implant, which is 2 millimeters thick and 8 millimeters wide, contains iron-oxide particles coated with antibodies that target a specific biomarker. A semi-permeable membrane allows the protein targets to enter the device, where they bind to the antibodies. In this study, the researchers implanted six sensors — two for each heart-attack biomarker — under the skin of each mouse, and read them using magnetic resonance imaging (MRI).
One important finding is that all three sensors’ output was shown to be proportional to the size of the damage to the heart, says Huang. Thus, not only can they potentially be used to detect a heart attack, but they could yield some quick information on its severity.
This study marks the first time anyone has used implantable sensors to detect three different biomarkers, says Lee Josephson, associate professor at MGH’s Center for Molecular Imaging Research. “This shows how generalizable this technique is,” says Josephson, who was not involved in this study. Potential applications include not only detecting heart disease and cancer, but also tracking glucose levels in diabetic patients, he says.
Cima is now developing an implant that measures pH (acidity level), which could be useful for detecting heart disease or cancer. (Tumors are more acidic than healthy tissue, and dramatic increase in acidity is a near-instantaneous indicator of heart attack.)
In the future, he hopes to modify these sensors to detect low levels of hard-to-detect bacteria or viruses, or migrating tumor cells. “This may be a way to look for extremely small, or extremely transient concentrations of biological markers,” he says.
The current version of the implant is usable for about two months, but Cima believes the devices could be made to last longer by using antibodies that don’t break down as quickly.