A protein that normally helps defend cells from infection can play a critical role in the development of lung cancer, according to MIT cancer biologists.
Their findings suggest that the protein, NF-kappaB, could be a promising target for new drugs against lung cancer, which kills more than one million people each year.
NF-kappaB is a transcription factor protein that is normally activated when a cell is under attack from a virus or bacterium. Previous studies have suggested that constant activation of NF-kappaB enhances survival of tumor cells.
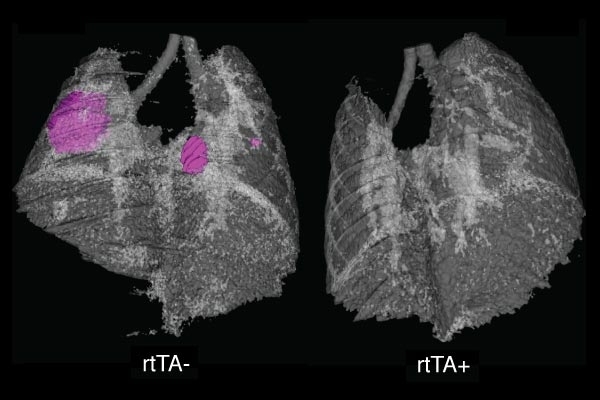
In the new study, reported in the Oct. 22 issue of Nature, the MIT team found that a particular pair of genetic circumstances is required to activate NF-kappaB in mouse lung tumors: expression of the cancer gene ras, and loss of the tumor suppressor gene p53. They also showed that inhibition of NF-kappaB in mice with that genetic profile can slow tumor growth.
In human lung cancer patients, the ras gene is active in 30 percent of patients, and p53 is lost in about 50 percent of tumors, meaning that about 15 percent overall have this combination. Drugs that inhibit NF-kappaB could potentially help treat such tumors, says Etienne Meylan, lead author of the paper and a postdoctoral associate in the laboratory of Tyler Jacks, director of the David H. Koch Institute for Integrative Cancer Research at MIT.
The researchers found that when they inhibited NF-kappaB in lung tumors of mice with this genetic profile, tumor growth slowed dramatically. During the three-week period following NF-kappaB inhibition, tumors in treated mice grew, on average, half as much as tumors in untreated mice. In some treated mice, tumors shrank.
Cancer biologists have been pursuing possible treatments based on NF-kappaB inhibition over the past decade or so, but during the past few years, a handful of studies have shown that inhibiting NF-kappaB can have adverse side effects. Because of that, "some people began to re-evaluate the potential of NF-kappaB as a target," says cancer researcher Sankar Ghosh of Columbia University Medical Center, who was not involved in the research. However, he says the new findings should jump start new efforts to target NF-kappaB in tumors with this specific genetic profile. "This paper shows the effects in such a dramatic and striking way, I think it will re-energize people who are interested in NF-kappaB as a potential target," says Ghosh.
The mouse model used in this study could be useful in those efforts, says Meylan. Additionally, "we could use this model to find better compounds — those that are more selective or more powerful," he says.
Their findings suggest that the protein, NF-kappaB, could be a promising target for new drugs against lung cancer, which kills more than one million people each year.
NF-kappaB is a transcription factor protein that is normally activated when a cell is under attack from a virus or bacterium. Previous studies have suggested that constant activation of NF-kappaB enhances survival of tumor cells.
In the new study, reported in the Oct. 22 issue of Nature, the MIT team found that a particular pair of genetic circumstances is required to activate NF-kappaB in mouse lung tumors: expression of the cancer gene ras, and loss of the tumor suppressor gene p53. They also showed that inhibition of NF-kappaB in mice with that genetic profile can slow tumor growth.
In human lung cancer patients, the ras gene is active in 30 percent of patients, and p53 is lost in about 50 percent of tumors, meaning that about 15 percent overall have this combination. Drugs that inhibit NF-kappaB could potentially help treat such tumors, says Etienne Meylan, lead author of the paper and a postdoctoral associate in the laboratory of Tyler Jacks, director of the David H. Koch Institute for Integrative Cancer Research at MIT.
The researchers found that when they inhibited NF-kappaB in lung tumors of mice with this genetic profile, tumor growth slowed dramatically. During the three-week period following NF-kappaB inhibition, tumors in treated mice grew, on average, half as much as tumors in untreated mice. In some treated mice, tumors shrank.
Cancer biologists have been pursuing possible treatments based on NF-kappaB inhibition over the past decade or so, but during the past few years, a handful of studies have shown that inhibiting NF-kappaB can have adverse side effects. Because of that, "some people began to re-evaluate the potential of NF-kappaB as a target," says cancer researcher Sankar Ghosh of Columbia University Medical Center, who was not involved in the research. However, he says the new findings should jump start new efforts to target NF-kappaB in tumors with this specific genetic profile. "This paper shows the effects in such a dramatic and striking way, I think it will re-energize people who are interested in NF-kappaB as a potential target," says Ghosh.
The mouse model used in this study could be useful in those efforts, says Meylan. Additionally, "we could use this model to find better compounds — those that are more selective or more powerful," he says.