As Jesse Patterson, an MIT research scientist, and Frank Lovell, a finance industry retiree with a penchant for travel, chatted in the Koch Institute auditorium after a public lecture, they realized the anomaly of the experience: Cancer patients rarely get to meet researchers working on their treatments, and cancer researchers rarely get to put a name and a face to the people they aim to help through their work.
Lovell was participating in a clinical trial for a prostate cancer therapy that combines the widely-used targeted therapy abiraterone with the Plk1 inhibitor onvansertib. Patterson, working in the laboratory of Professor Michael Yaffe, the David H. Koch Professor of Science and director of the MIT Center for Precision Cancer Medicine, played a significant role in identifying the new drug combination and its powerful potential.
While their encounter was indeed fortunate, it was not random. They never would have met if not for the human synergy showcased at that evening’s SOLUTIONS with/in/sight event, the result of collaborative relationships built between research labs, clinical centers, and industry. Patterson and Yaffe were on hand to tell the story of the science behind their new drug combination, and were joined by some of the partners who helped translate their results into a clinical trial: David Einstein, clinical oncologist at Beth Israel Deaconess Medical Center, and Mark Erlander, chief scientific officer of Trovagene Oncology, the biotech company that developed onvansertib.
Network synergy
The need for new prostate cancer therapies is acute. Prostate cancer is the leading diagnosis among men for non-skin cancer and the second-leading cancer killer among men in the United States. Abiraterone works by shutting off androgen synthesis and interfering with the androgen receptor pathway, which plays a crucial role in prostate cancer cells’ ability to survive and divide. However, cancer cells eventually evolve resistance to abiraterone. New, more powerful drug combinations are needed to circumvent or delay the development of resistance.
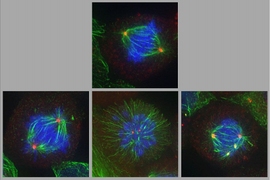
Patterson and his colleagues in the Yaffe lab hypothesized that by targeting both the androgen receptor and other pathways critical to cancer cell proliferation, they could produce a synergistic effect — that is, a combination effect that is much greater than the sum of each drug’s effect by itself. Plk1, a pathway critical to each stage of cell division, was of longstanding interest to the Yaffe group, and was among those Patterson strategically selected for investigation as a potential partner target for androgen receptor. In screens of prostate cancer cell lines and in xenograft tumors, the researchers found that abiraterone and Plk1 inhibitors both interfere with cell division when delivered singly, but that together, those effects are amplified and far more often lethal to cancer cells.
An unexpected phone call from Mark Erlander at Trovagene, a San Diego-based clinical-stage biotech company, was instrumental in translating the Yaffe Lab’s research results into clinical trials.
Erlander had learned that MIT held a patent for the combination of Plk1 inhibitors and anti-androgens for any cancer — the result of Yaffe Lab studies. Although he did not know Yaffe personally and lived a continent away, Erlander picked up the phone and invited Yaffe for coffee. “This was worth flying across the country,” Erlander said.
Still in scrubs, Yaffe, who is an attending surgeon at Beth Israel Deaconess Medical Center in addition to his academic roles, chatted with Erlander during his shift break at the hospital. The new collaboration was on its way.
Speaking Frankly
While Erlander had the Plk1 inhibitor and the Yaffe Lab had the science behind it, they were still missing an important component of any clinical trial: patients. Yaffe enlisted doctors David Einstein and Steven Balk, both at Beth Israel Deaconess Medical Center and Dana Farber/Harvard Cancer Center, with whom he had worked on related research supported by the Bridge Project, to bring clinical translation expertise and patient access.
By the time clinical trials began in 2019, Frank Lovell was ready for a new treatment. When his prostate cancer was first diagnosed about a decade ago, he was treated with surgery and radiation. When the cancer came back five years later, he received a hormonal treatment that stopped working within three years. He started to see Einstein, an oncologist who specialized in novel therapies, and tried yet another treatment, this one losing effectiveness after a year. Then he joined Einstein’s trial.
For Lovell, the new combination of drugs was “effective in a wonderful way.” Many of the patients in the trial — 72 percent of those who completed phase 2 — showed declining or stabilized levels of prostate-specific androgen (PSA), indicating a positive response to the treatment. Lovell’s PSA levels stabilized, too, and he reports that he experienced very few side effects.
But most importantly, noted Lovell, “I say thank you to Dr. Einstein, Dr. Patterson, and Dr. Yaffe. They brought me hope and time.”
The gratitude is mutual.
“I especially want to thank Frank and all the patients like him who have volunteered to be on these clinical trials,” says Yaffe. “Without patients like Frank, we would never know how to better treat these types of cancers.”
Lovell is no longer in the trial for now, but enjoying making his rounds from Cape Cod in the summer; to Paris and Cannes, France, and then Hawaii in the autumn; and to Naples, Florida, in the winter, on top of visiting with family and a wide circle of friends. “Illness has not stopped me from living a normal life,” Lovell said. “You wouldn’t think I was sick.”
Meanwhile, Yaffe, Patterson, and their research collaborators are still at work. They are optimizing drug delivery regimens to maximize the time on treatment and minimize toxicity, as well as finding biomarkers that help identify which patients will best respond to the combination. They are also looking to understand the mechanism behind the synergy better, which in turn may help them find more effective partners for onvansertib, and to identify other cancer types, such as ovarian cancer, for which the combination may be effective.