Editor’s note: This is part of a series of articles linking the work of MIT’s emeritus faculty members with the current state of research in their given fields.
When MIT chemical engineering professor Paula Hammond cleans out her lab every few years, her students occasionally find bottles of the chemical polyethylene oxide that date back to the 1980s. The bottles are relics of the previous lab occupant, Professor Emeritus Edward Merrill, and a reminder of his legacy in developing polymers for biomedical uses.
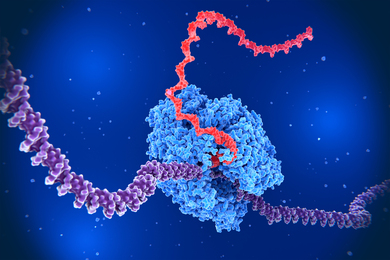
Merrill, one of a handful of researchers who pioneered the field of biomedical engineering in the 1960s and 70s, showed that polyethylene oxide, a polymer (long chain) of repeating units called ethers, is remarkably inert when in contact with blood, whereas most other materials cause blood to clot upon contact. He suspected this quality would make it ideal for biomedical applications such as drug delivery.
Today, polyethylene oxide is ubiquitous in the biomedical and pharmaceutical industries, and Hammond and others are developing the polymer into nanoparticles for cancer-drug delivery. Hammond’s “time bomb” nanoparticles can flow through the bloodstream without being absorbed by cells, until they reach a tumor and release their chemotherapy payload.
Polyethylene oxide, also known as polyethylene glycol (PEG), “turns out to be one of the most important, critical elements of designing the polymeric systems we believe can help us in targeting drugs for cancer,” says Hammond, the Bayer Professor of Chemical Engineering.
During his 60-year career at MIT, Merrill also used his expertise in polymer chemistry, especially the study of membranes, to make major contributions to the development of the artificial kidney and oxygenation of the blood during open-heart surgery.
‘Sticky stuff’
Although Merrill’s father was a chemical engineer, he was at first reluctant to follow in his father’s footsteps. At Harvard, he majored in chemistry and classics. His senior year, he planned to take a course in advanced physical chemistry, but wandered into the wrong classroom and ended up in a chemical engineering course taught by MIT Professor William Henry McAdams. “It kind of intrigued me, so instead of going to the correct classroom, I stayed on with it and did reasonably well in it,” recalls Merrill, now 86.
McAdams encouraged Merrill to come to MIT for graduate school, where he earned his PhD in chemical engineering in 1947. He did his thesis in polymer chemistry, an area he hadn’t encountered at Harvard. “It was an ignominious pursuit. Pure chemists would not touch it. It was sticky stuff, gooey stuff,” Merrill says.
Merrill then spent three years working at the Dewey & Almy Chemical Company, where he studied rheology — the flow of polymer materials, such as rubber cement. He returned to MIT for good in 1950, joining the faculty as an assistant professor.
Because of his experience in rheology, researchers at Brigham and Women’s Hospital asked him to help them measure the viscosity of blood, which is how he got into biomedical engineering, around 1960. That work led him to devise a better way to oxygenate blood during open-heart surgery, using membranes permeable to oxygen and carbon dioxide. He also developed membranes to filter blood for artificial kidney machines.
At the time, only a handful of engineers around the United States were delving into biomedicine. Merrill’s colleagues told him that his work in blood rheology might undermine his shot at tenure. But the risk paid off, and Merrill went on to launch biomedical engineering as a major focus of MIT’s chemical engineering department, influencing hundreds if not thousands of MIT students.
“While it may be common to have a medical focus in chemical engineering curricula now, it sure wasn’t in 1957, or even when I came to MIT in 1970,” Robert Langer, MIT Institute Professor, said at a May 14 symposium honoring Merrill’s 60 years on the MIT faculty. “When you look at the people he’s touched and the people they’ve touched, it’s truly remarkable. It’s hard for me to think of anyone who’s had a greater impact.”
The Cheshire Cat
Hammond first met Merrill as a senior at MIT in 1983, when she took his class in polymer chemistry. She appreciated Merrill’s focus on chemistry at the molecular level, which was a change from the usual chemical engineering focus on materials as a continuum.
“As an undergrad, the reason I wanted to become a chemical engineer was because I loved chemistry,” Hammond recalls. “When I started to take my chemical engineering courses I missed the sense that something molecular was going on. I loved chemistry and I wanted to see it again. So I took as an elective one of Professor Merrill’s classes in polymers and just got fascinated.”
In 1988, Hammond returned to MIT to get her PhD in chemical engineering and took two more polymer chemistry classes with Merrill. “Physical chemistry of polymers can be particularly difficult to conceptualize but he was able to make these concepts incredibly clear and fascinating at the same time,” she says.
Recognizing the difficulty of the material he was teaching, especially thermodynamics of polymers, Merrill often used an analogy from one of his favorite works, “Alice in Wonderland,” to hearten his students. In the story, the Cheshire Cat often climbs up a tree and then slowly disappears, leaving only the grinning mouth.
“Some of these things are very difficult concepts. It takes a lot of thinking, a lot of soaking in, before you can get it,” Merrill says. “I said to the students, this is going to be like the Cheshire Cat’s grin. You’re going to go out of the room and you’ll have this vague idea that you saw something, but the rest of the idea will be gone.”
The students eventually got it, though, says Hammond, who co-taught two courses with Merrill in the late 1990s, soon after she joined the MIT faculty. “It was really amazing to be in the classroom with him and to learn how he engages the students,” she says. When they began teaching together, Hammond asked Merrill for his course notes, which turned out to be three or four sheets stapled together. “I remember thinking, ‘Oh my God, he does all of this just streaming from his head,’” says Hammond. “It amazed me.”
Throughout his career, Merrill, who officially retired from MIT in 2001, made it a point to keep up with what was happening in industrial chemical engineering, and to convey that aspect of the field to his students. He often designed his labs to incorporate biomaterials such as bone cement, used to anchor artificial joints. Because of that exposure to real-world applications, “we end up with chemical engineers coming out of our department with this perspective that I think is unique,” says Hammond.
When MIT chemical engineering professor Paula Hammond cleans out her lab every few years, her students occasionally find bottles of the chemical polyethylene oxide that date back to the 1980s. The bottles are relics of the previous lab occupant, Professor Emeritus Edward Merrill, and a reminder of his legacy in developing polymers for biomedical uses.
Merrill, one of a handful of researchers who pioneered the field of biomedical engineering in the 1960s and 70s, showed that polyethylene oxide, a polymer (long chain) of repeating units called ethers, is remarkably inert when in contact with blood, whereas most other materials cause blood to clot upon contact. He suspected this quality would make it ideal for biomedical applications such as drug delivery.
Today, polyethylene oxide is ubiquitous in the biomedical and pharmaceutical industries, and Hammond and others are developing the polymer into nanoparticles for cancer-drug delivery. Hammond’s “time bomb” nanoparticles can flow through the bloodstream without being absorbed by cells, until they reach a tumor and release their chemotherapy payload.
Polyethylene oxide, also known as polyethylene glycol (PEG), “turns out to be one of the most important, critical elements of designing the polymeric systems we believe can help us in targeting drugs for cancer,” says Hammond, the Bayer Professor of Chemical Engineering.
During his 60-year career at MIT, Merrill also used his expertise in polymer chemistry, especially the study of membranes, to make major contributions to the development of the artificial kidney and oxygenation of the blood during open-heart surgery.
‘Sticky stuff’
Although Merrill’s father was a chemical engineer, he was at first reluctant to follow in his father’s footsteps. At Harvard, he majored in chemistry and classics. His senior year, he planned to take a course in advanced physical chemistry, but wandered into the wrong classroom and ended up in a chemical engineering course taught by MIT Professor William Henry McAdams. “It kind of intrigued me, so instead of going to the correct classroom, I stayed on with it and did reasonably well in it,” recalls Merrill, now 86.
McAdams encouraged Merrill to come to MIT for graduate school, where he earned his PhD in chemical engineering in 1947. He did his thesis in polymer chemistry, an area he hadn’t encountered at Harvard. “It was an ignominious pursuit. Pure chemists would not touch it. It was sticky stuff, gooey stuff,” Merrill says.
Merrill then spent three years working at the Dewey & Almy Chemical Company, where he studied rheology — the flow of polymer materials, such as rubber cement. He returned to MIT for good in 1950, joining the faculty as an assistant professor.
Because of his experience in rheology, researchers at Brigham and Women’s Hospital asked him to help them measure the viscosity of blood, which is how he got into biomedical engineering, around 1960. That work led him to devise a better way to oxygenate blood during open-heart surgery, using membranes permeable to oxygen and carbon dioxide. He also developed membranes to filter blood for artificial kidney machines.
At the time, only a handful of engineers around the United States were delving into biomedicine. Merrill’s colleagues told him that his work in blood rheology might undermine his shot at tenure. But the risk paid off, and Merrill went on to launch biomedical engineering as a major focus of MIT’s chemical engineering department, influencing hundreds if not thousands of MIT students.
“While it may be common to have a medical focus in chemical engineering curricula now, it sure wasn’t in 1957, or even when I came to MIT in 1970,” Robert Langer, MIT Institute Professor, said at a May 14 symposium honoring Merrill’s 60 years on the MIT faculty. “When you look at the people he’s touched and the people they’ve touched, it’s truly remarkable. It’s hard for me to think of anyone who’s had a greater impact.”
The Cheshire Cat
Hammond first met Merrill as a senior at MIT in 1983, when she took his class in polymer chemistry. She appreciated Merrill’s focus on chemistry at the molecular level, which was a change from the usual chemical engineering focus on materials as a continuum.
“As an undergrad, the reason I wanted to become a chemical engineer was because I loved chemistry,” Hammond recalls. “When I started to take my chemical engineering courses I missed the sense that something molecular was going on. I loved chemistry and I wanted to see it again. So I took as an elective one of Professor Merrill’s classes in polymers and just got fascinated.”
In 1988, Hammond returned to MIT to get her PhD in chemical engineering and took two more polymer chemistry classes with Merrill. “Physical chemistry of polymers can be particularly difficult to conceptualize but he was able to make these concepts incredibly clear and fascinating at the same time,” she says.
Recognizing the difficulty of the material he was teaching, especially thermodynamics of polymers, Merrill often used an analogy from one of his favorite works, “Alice in Wonderland,” to hearten his students. In the story, the Cheshire Cat often climbs up a tree and then slowly disappears, leaving only the grinning mouth.
“Some of these things are very difficult concepts. It takes a lot of thinking, a lot of soaking in, before you can get it,” Merrill says. “I said to the students, this is going to be like the Cheshire Cat’s grin. You’re going to go out of the room and you’ll have this vague idea that you saw something, but the rest of the idea will be gone.”
The students eventually got it, though, says Hammond, who co-taught two courses with Merrill in the late 1990s, soon after she joined the MIT faculty. “It was really amazing to be in the classroom with him and to learn how he engages the students,” she says. When they began teaching together, Hammond asked Merrill for his course notes, which turned out to be three or four sheets stapled together. “I remember thinking, ‘Oh my God, he does all of this just streaming from his head,’” says Hammond. “It amazed me.”
Throughout his career, Merrill, who officially retired from MIT in 2001, made it a point to keep up with what was happening in industrial chemical engineering, and to convey that aspect of the field to his students. He often designed his labs to incorporate biomaterials such as bone cement, used to anchor artificial joints. Because of that exposure to real-world applications, “we end up with chemical engineers coming out of our department with this perspective that I think is unique,” says Hammond.