It’s well established that critical human body functions, including sleep, hormone production and regulation of body temperature, follow a circadian (24-hour) cycle. These genetically programmed patterns stay in effect even under isolation from the naturally occurring daily light-dark cycles of the sun, and are found in nearly all organisms — animals, plants, fungi and even some bacteria.
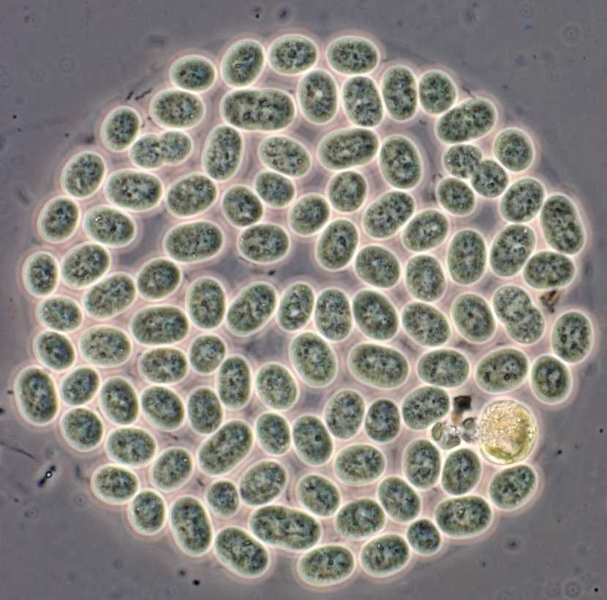
Although bacteria don’t “sleep” in the same way humans do, a type of bacteria known as cyanobacteria undergoes daily cycles of activity and rest. These bacteria depend on sunlight to perform photosynthesis, so they have evolved to be most active in daylight.
A new study of cyanobacteria by a team of researchers at MIT and the University of California at San Diego has revealed, for the first time, how those circadian rhythms control the bacteria’s rate of cell division (their method of reproduction) in single cells. “These cells have to keep dividing, and the circadian oscillator regulates when they divide,” says Bernardo Pando, an MIT graduate student in physics and one of the lead authors of a paper describing the findings in the March 18 online edition of Science.
In multicellular animals, including humans, cell division is critical for renewal and repair, while out-of-control cell division leads to cancer, so “understanding how cells are dividing is really of fundamental importance,” says Susan Golden, professor of molecular biology at the University of California at San Diego and an author of the paper.
Divide, rest, repeat
In 1998, Golden and other researchers identified three so-called “clock proteins” that control circadian rhythms in cyanobacteria. There has been some evidence that the circadian clock regulates cell division, but the exact relationship was unclear.
The MIT-UCSD team, led by MIT biophysics professor Alexander van Oudenaarden, found that under conditions of moderate constant light, the cyanobacteria undergo cell division about once per day, and the divisions take place mostly at the midpoint of the 24-hour cycle.
The researchers then sped up the cell cycle by boosting the intensity of light. That enabled the cells to photosynthesize more, increasing the amount of energy available to them. The cells did divide more frequently, but in a pattern still linked to the circadian clock — they divided a quarter of the way into the cycle, and again three-quarters into the cycle.
The team also showed that in all conditions, the cyanobacteria enter a resting phase about 19 hours into the circadian cycle, after which they will not divide until the next cycle begins.
Key to the new finding is a technique the researchers devised to study circadian rhythms in single cells, says van Oudenaarden, the senior author of the paper. The team tracked individual cells over a week. Proteins that govern the circadian clock were tagged with yellow fluorescent protein, so each cell’s position in the 24-hour cycle could be pinpointed. Cells were also photographed every 40 minutes, so researchers could see when they divided.
The single-cell tracking technique could also be used to reveal linkages between the circadian clock and other cyclical cell process, such as metabolism. “The circadian cycle has to ride herd on all these other cycles and make sure they stay in synchrony and that cell activity is not chaotic,” says Golden.
Golden is planning follow-up studies in cyanobacteria, but yeast and mammalian cells could also be logical targets for such studies, says van Oudenaarden.
In a recent paper published in the journal Cell, van Oudenaarden and Golden reported the molecular mechanism of how clock proteins control the cell cycle in cyanobacteria. The proteins (KaiA, KaiB and KaiC) control the action of another protein called FtsZ, preventing it from going to the middle of the cell and forming a ring necessary for cell division.
Taken together, the papers are “excellent work connecting cell division to the circadian clock,” says Paul Hardin, a biologist at Texas A&M University who studies circadian rhythms in fruit flies.
Although bacteria don’t “sleep” in the same way humans do, a type of bacteria known as cyanobacteria undergoes daily cycles of activity and rest. These bacteria depend on sunlight to perform photosynthesis, so they have evolved to be most active in daylight.
A new study of cyanobacteria by a team of researchers at MIT and the University of California at San Diego has revealed, for the first time, how those circadian rhythms control the bacteria’s rate of cell division (their method of reproduction) in single cells. “These cells have to keep dividing, and the circadian oscillator regulates when they divide,” says Bernardo Pando, an MIT graduate student in physics and one of the lead authors of a paper describing the findings in the March 18 online edition of Science.
In multicellular animals, including humans, cell division is critical for renewal and repair, while out-of-control cell division leads to cancer, so “understanding how cells are dividing is really of fundamental importance,” says Susan Golden, professor of molecular biology at the University of California at San Diego and an author of the paper.
Divide, rest, repeat
In 1998, Golden and other researchers identified three so-called “clock proteins” that control circadian rhythms in cyanobacteria. There has been some evidence that the circadian clock regulates cell division, but the exact relationship was unclear.
The MIT-UCSD team, led by MIT biophysics professor Alexander van Oudenaarden, found that under conditions of moderate constant light, the cyanobacteria undergo cell division about once per day, and the divisions take place mostly at the midpoint of the 24-hour cycle.
The researchers then sped up the cell cycle by boosting the intensity of light. That enabled the cells to photosynthesize more, increasing the amount of energy available to them. The cells did divide more frequently, but in a pattern still linked to the circadian clock — they divided a quarter of the way into the cycle, and again three-quarters into the cycle.
The team also showed that in all conditions, the cyanobacteria enter a resting phase about 19 hours into the circadian cycle, after which they will not divide until the next cycle begins.
Key to the new finding is a technique the researchers devised to study circadian rhythms in single cells, says van Oudenaarden, the senior author of the paper. The team tracked individual cells over a week. Proteins that govern the circadian clock were tagged with yellow fluorescent protein, so each cell’s position in the 24-hour cycle could be pinpointed. Cells were also photographed every 40 minutes, so researchers could see when they divided.
The single-cell tracking technique could also be used to reveal linkages between the circadian clock and other cyclical cell process, such as metabolism. “The circadian cycle has to ride herd on all these other cycles and make sure they stay in synchrony and that cell activity is not chaotic,” says Golden.
Golden is planning follow-up studies in cyanobacteria, but yeast and mammalian cells could also be logical targets for such studies, says van Oudenaarden.
In a recent paper published in the journal Cell, van Oudenaarden and Golden reported the molecular mechanism of how clock proteins control the cell cycle in cyanobacteria. The proteins (KaiA, KaiB and KaiC) control the action of another protein called FtsZ, preventing it from going to the middle of the cell and forming a ring necessary for cell division.
Taken together, the papers are “excellent work connecting cell division to the circadian clock,” says Paul Hardin, a biologist at Texas A&M University who studies circadian rhythms in fruit flies.