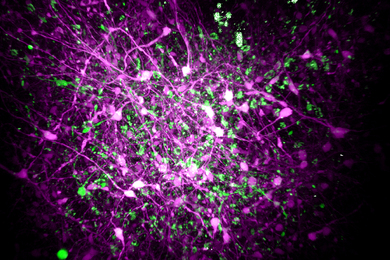
Few conditions are more detrimental to human brains than the one popularly referred to as mad cow disease. But now there's reason to suspect that the protein which, when malformed, causes bovine spongiform encephalopathy in cows and Creutzfeldt-Jakob disease in people, might also be necessary for healthy brain function.
Researchers from MIT, the Whitehead Institute for Biomedical Research and Harvard Medical School/Massachusetts General Hospital have discovered that the normal form of this detrimental protein may actually help the brain create neurons, those electricity-conducting cells that make cognition possible.
"It's been difficult to understand why this prion protein, which when malformed subjects us to this horrible disease, is so abundant in our brains in the first place," says Whitehead member Susan Lindquist, who is also a professor of biology at MIT. "We've known for years what happens when this protein goes wrong. Now we're starting to see what its normal form does right."
Along with Jeffrey Macklis of Harvard Medical School and Massachusetts General Hospital, Lindquist is co-senior author on a paper published this week in the Proceedings of the National Academy of Sciences.
For more than 10 years, researchers have known that a protein called PrP causes mad cow disease and its human equivalent, Creutzfeldt-Jakob disease, when it forms incorrectly. PrP is a prion, a class of proteins that has the unusual ability to recruit other proteins to change their shape. (PrP is shorthand for "prion protein".)
This is significant because a protein's form determines its function. When a prion changes shape, or "misfolds," it creates a cascade of neighboring proteins all assuming that particular conformation. In some organisms, such as yeast cells, this process can be harmless or even beneficial. But in mammals, it can lead to the fatal brain lesions that characterize diseases such as Creutzfeldt-Jakob.
Curiously, however, PrP can be found throughout healthy human bodies, particularly in the brain. In fact, it's found in many mammalian species, and only on the rarest occasions does it misfold and cause disease. Clearly, scientists have reasoned, such a widely conserved protein also must play a beneficial role.
In 1993, scientists created a line of mice in which the gene that codes for PrP was knocked out, preventing the mice from expressing the prion in any tissues. Surprisingly, the mice showed no sign of any ill effect. The only difference between these mice and the control mice was that the animals in which the gene was knocked out were incapable of contracting prion-related neurodegenerative disease when infected. Researchers knew then that PrP was necessary for mad-cow type diseases; any other kind of normal function remained unknown.
Recently, researchers from the labs of Lindquist and Whitehead member and MIT biology Professor Harvey Lodish discovered that PrP helps preserve stem cells in the blood. Because of this, Lindquist teamed up with Macklis to see if there might also be a similar connection between PrP and cells in the brain, where the prion protein is far more abundant.
Andrew Steele, a graduate student from the Lindquist lab, teamed up with Jason Emsley and Hande Ozdinler, postdoctoral researchers in the Macklis lab, to investigate the effects PrP might have on neurogenesis. (Neurogenesis is the process by which the brain creates new neurons in the developing embryonic brain and, to a limited extent, in the adult brain.) To do this they studied embryonic brain tissue from three kinds of mice: those in which the PrP gene was permanently disabled, or knocked out; those in which the gene was over-expressed, producing an unusually large amount of PrP; and normal control mice.
Steele and Emsley isolated neural precursor cells -- early-stage cells that give rise to mature neurons and so-called glial support cells. After placing these embryonic precursor cells under culture conditions that enabled them to grow and differentiate, they noticed striking differences. Cells from the knock-out mice remained in the precursor stage for a long time, compared to the control mice. But cells in which PrP was overexpressed began forming mature neurons almost immediately.
"The more PrP you have, the faster you become a neuron. The less you have, the longer you'll stay in a precursor state," says Steele.
This research was funded by the National Institutes of Health/National Institute of Neurological Disorders and Stroke, the Ellison Medical Research Foundation, Paralyzed Veterans of America/Travis Roy Foundation, the Children's Neurobiological Solutions Foundation, the Heart and Stroke Foundation of Canada and the Harvard Center for Neurodegeneration and Repair.
A version of this article appeared in MIT Tech Talk on March 15, 2006 (download PDF).