A significant challenge for researchers in materials science and drug discovery is that even the most minor change to a molecule’s structure can completely alter its function. Historically, making these adjustments meant researchers had to re-synthesize the target molecule from scratch — a time-consuming and expensive bottleneck akin to tearing down a house just to move a lamp.
In an exciting discovery recently published in Nature, MIT chemists led by Professor Alison Wendlandt have developed a precision technique that allows scientists to seamlessly relocate alcohol functional groups from one spot on a molecule to a neighboring site. This process bypasses the need to rebuild the entire structure and is the result of a multi-year collaboration with Bristol Myers Squibb.
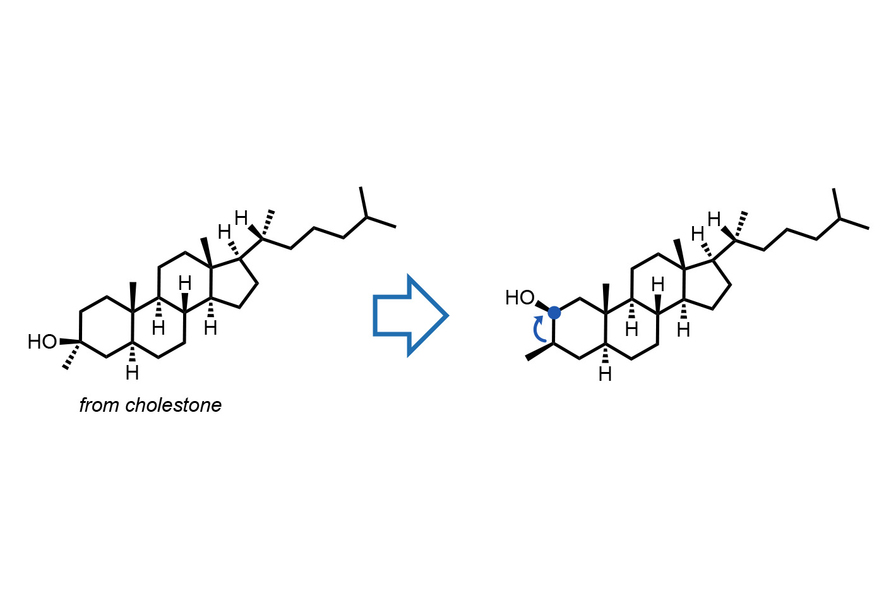
Functional group repositioning
Using a special light-sensitive molecule called decatungstate as a catalyst, the reaction triggers a highly controlled “migration” of the alcohol group. The process is remarkably predictable, ensuring the molecule retains its precise 3D shape and orientation throughout the move.
The ability to implement subtle structural tweaks without the waste of “from-scratch” synthesis eliminates a primary hurdle that has long plagued the field. Furthermore, because the reaction is gentle enough to work on complex, nearly finished structures, it serves as a powerful fine-tuning tool for late-stage drug candidates.
Precision editing to unlock new chemical designs
When combined with existing chemical methods, this tool provides new pathways to create challenging molecular architectures and oxygenation patterns that were previously out of reach.
“This alcohol migration strategy allows for precise, molecular-level tuning of oxygen atom positions,” says Qian Xu, the co-first author of the paper and a postdoc in the Wendlandt Group. “With predictable stereo- and regioselectivity and late-stage operability, it presents an enticing chance to modify natural products and drug molecules through ‘editing.’”
Ultimately, this precision editing tool holds the potential to dramatically improve the efficiency of molecular design campaigns, accelerating the development of new pharmaceuticals, materials, and agrochemicals.
In addition to Wendlandt and Xu, MIT contributors include co-lead author and graduate student Yichen Nie, recent postdoc Ronghua Zhang, and professor of chemistry Jeremiah A. Johnson. Other authors include Jacob-Jan Haaksma of the University of Groningen in The Netherlands; Natalie Holmberg-Douglas, Farid van der Mei, and Chloe Williams of of Bristol Myers Squibb; and Paul M. Scola of Actithera.