Doctors warn that marijuana use during pregnancy may have harmful effects on the development of a fetus, in part because the cannabinoid receptors activated by the drug are known be critical for enabling a developing brain to wire up properly. Now, scientists at MIT’s McGovern Institute for Brain Research have learned that cannabinoid receptors’ critical role in brain development does not end at birth.
In today's online issue of eNeuro, scientists led by McGovern investigator Ann Graybiel report that mice need the cannabinoid receptor CB1R to establish connections within the brain’s dopamine system that take shape soon after birth. The finding raises concern that marijuana use by nursing moms, who pass the CB1R-activating compound THC to their infants when they breastfeed, might interfere with brain development by disrupting cannabinoid signaling.
“This is a real change to one of the truly important systems in the brain — a major controller of our dopamine,” says Graybiel, who is an Institute Professor and a faculty member in the Department of Brain and Cognitive Sciences. Dopamine exerts a powerful influence over our motivations and behavior, and changes to the dopamine system contribute to disorders from Parkinson’s disease to addiction. Thus, the researchers say, it is vital to understand whether postnatal drug exposure might put developing dopamine circuits at risk.
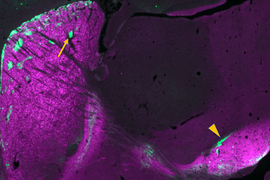
Cannabinoid receptors in the brain are important mediators of mood, memory, and pain. Graybiel’s lab became interested in CB1R due to their dysregulation in Huntington’s and Parkinson’s diseases, both of which impair the brain’s ability to control movement and other functions. While investigating the receptor’s distribution in the brain, they discovered that in the adult mice, CB1R is abundant within small compartments within the striatum called striosomes. The receptor was particularly concentrated within the neurons that connect striosomes to a dopamine-rich area of the brain called the substantia nigra, via structures that Graybiel’s team has dubbed striosome-dendron bouquets.
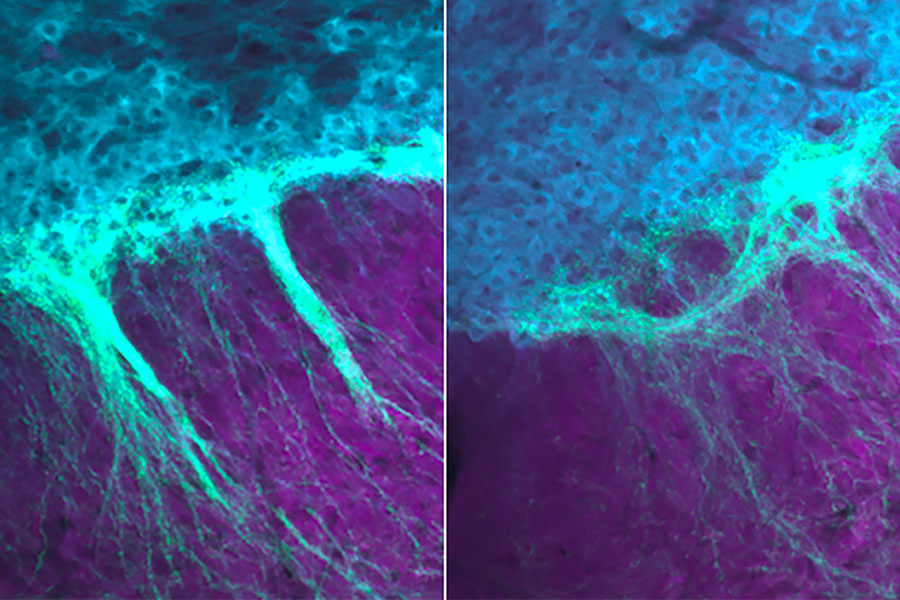
Striosome-dendron bouquets are easy to overlook within the densely connected network of the brain. But when the cells that make up the bouquets are labeled with a fluorescent protein, the bouquets become visible — and their appearance is striking, says Jill Crittenden, a research scientist in Graybiel’s lab.
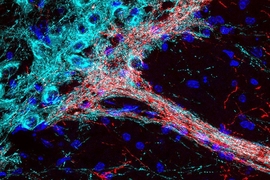
Striosomal neurons form these bouquets by reaching into the substantia nigra, whose cells use dopamine to influence movement, motivation, learning, and habit formation. Clusters of dopamine-producing neurons form dendrites there that intertwine tightly with incoming axons from the striosomal neurons. The resulting structures, whose intimately associated cells resemble the bundled stems of a floral bouquet, establish so many connections that they give striosomal neurons potent control over dopamine signaling.
By tracking the bouquets’ emergence in newborn mice, Graybiel’s team found that they form in the first week after birth, a period during which striosomal neurons are ramping up production of CB1R. Mice genetically engineered to lack CB1R, however, can’t make these elaborate but orderly bouquets. Without the receptor, fibers from striosomes extend into the substantia nigra, but fail to form the tightly intertwined “bouquet stems” that facilitate extensive connections with their targets. This disorganized structure is apparent as soon as bouquets arise in the brains of young pups and persists into adulthood. “There aren’t those beautiful, strong fibers anymore,” Crittenden says. “This suggests that those very strong controllers over the dopamine system function abnormally when you interfere with cannabinoid signaling.”
The finding was a surprise. Without zeroing in on striosome-dendron bouquets, it would be easy to miss CB1R’s impact on the dopamine system, Crittenden says. Plus, she adds, prior studies of the receptor’s role in development largely focused on fetal development. The new findings reveal that the cannabinoid system continues to guide the formation of brain circuits after birth.
Graybiel notes that funds from donors including the Broderick Fund for Phytocannabinoid Research at MIT, the Saks Kavanaugh Foundation, the Kristin R. Pressman and Jessica J. Pourian ‘13 Fund, Mr. Robert Buxton, and the William N. and Bernice E. Bumpus Foundation enabled her team’s studies of CB1R’s role in shaping striosome-dendron bouquets.
Now that they have shown that CB1R is needed for postnatal brain development, it will be important to determine the consequences of disrupting cannabinoid signaling during this critical period — including whether passing THC to a nursing baby impacts the brain’s dopamine system.