A study by MIT neuroscientists into how seemingly similar neuronal subtypes drive locomotion in the fruit fly reveals an unexpected diversity as the brain’s commands were relayed to muscle fibers. A sequence of experiments found a dramatic difference between the two nerve cells: One neuron scrambled to adjust to different changes by the other, but received no requital in response when circumstances were reversed.
The findings, published in the Journal of Neuroscience, suggest that these subclasses of neurons, which are also found abundantly in people and many other animals, exhibit a previously unappreciated diversity in their propensity to respond to changes, a key property known as “synaptic plasticity.” Synaptic plasticity is considered an essential mechanism of how learning and memory occur in the brain, and aberrations in the process are likely central to disorders such as autism.
“By seeing that these two different types of motor neurons actually show very distinct types of plasticity, that’s exciting because it means it’s not just one thing happening,” says senior author Troy Littleton, a member of The Picower Institute for Learning and Memory and the Menicon Professor of Neuroscience in MIT’s departments of Biology and of Brain and Cognitive Sciences. “There’s multiple types of things that can be altered to change connectivity within the neuromuscular system.”
Tonic and phasic neurons
Both of the neurons work in the same way, by emitting the neurotransmitter glutamate onto their connections, or synapses, with the muscles. But these two neurons do so with different styles. The “tonic” neuron, which connects only to a single muscle, emits its glutamate at a constant but low rate while the muscle is active. Meanwhile, the “phasic” neuron connects to a whole group of muscles and jumps in with a strong quick pulse of activity to spring the muscles into action.
Heading into the study, Littleton and lead author Nicole Aponte-Santiago PhD '20 were curious to explore whether these different neurons compete or cooperate to drive the muscle fibers, and if they exhibited different plasticity when their functions were altered. To get started on what ultimately became her dissertation research, Aponte-Santiago developed the means to tailor genetic alterations specifically in each of the two neurons.
“The reason we were able to answer these questions in the first place was because we produced tools to start differentially manipulating one neuron versus the other one, or label one versus the other one,” says Aponte-Santiago, who earned her PhD in Littleton’s lab earlier this spring and is now a postdoc at the University of California at San Francisco.
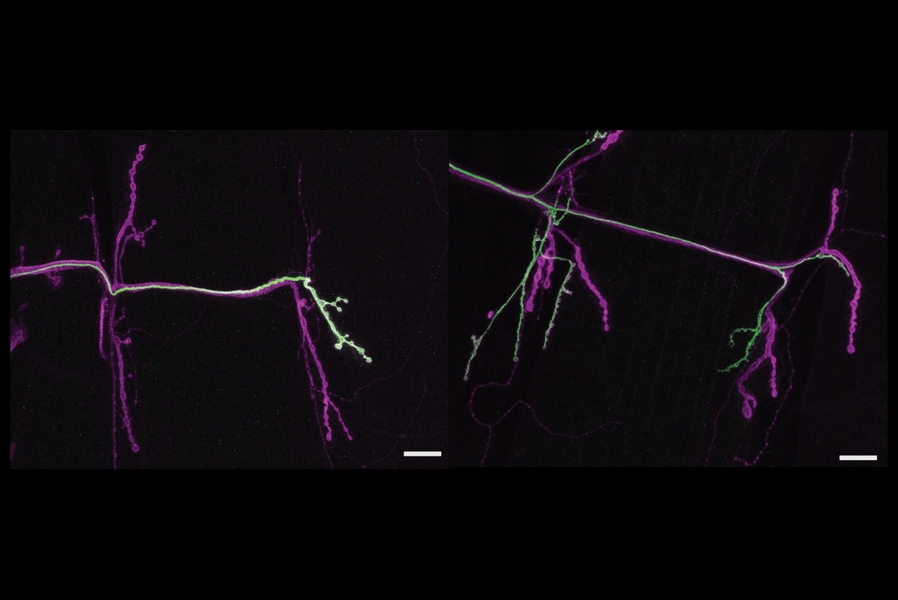
With genetic access to each neuron, Aponte-Santiago distinctly labeled them to watch each one grow in fly larvae as they developed. She saw that the tonic neuron reached the muscle first and that the phasic one connected to the muscle later. She also observed that unlike in mammals, the neurons did not compete to control the muscle but remained side by side, each contributing in its characteristic way to the total electrical activity needed to drive movement.
To study the neurons’ plasticity, Aponte-Santiago employed two manipulations of each neuron. She either wiped them out completely by making them express a lethal protein called “reaper,” or she substantially tamped down their glutamate activity via expression of tetanus toxin.
When she wiped out the phasic neuron with reaper, the tonic neuron quickly stepped up its signaling, attempting to compensate as much as it could. But in flies where she wiped out the tonic neuron, the phasic neuron didn’t budge at all, continuing as if nothing had changed.
Similarly, when Aponte-Santiago reduced the activity of the phasic neuron with the toxin, the tonic neuron increased the number of boutons and active zone structures in its synapses to respond to the loss of its partner. But when she reduced the activity of the tonic neuron, the phasic neuron again didn’t appear to respond.
In all the experiments, the muscle received less overall drive from the neurons than when everything was normal. And while the phasic neuron apparently didn’t bother to make up for any loss on the part of the tonic neuron, the tonic neuron employed different means to compensate — either increasing its signaling or by enhancing the number of its connections on the muscle — depending on how the phasic neuron was diminished.
“It was quite intriguing that Nicole found that when the phasic input wasn’t there, there was a unique form of plasticity that the tonic neuron showed,” Littleton says, “but if the phasic neuron was there and wasn’t working, the tonic neuron behaved in a very different way.”
Another intriguing aspect of the study is the role of the muscle itself, which may be an active intermediary of the plasticity, Littleton says. The neurons may not sense when each other have been wiped out or inactivated. Instead, the muscle appears to call for those changes.
“Even though a muscle has two distinct inputs, it can sort of uniquely control those two,” Littleton says. “When the muscle is getting glutamate, does it know whether it is coming from the tonic or the phasic neuron, and does it care? It appears that it does care, that it really needs the tonic more than the phasic. When the phasic is gone it shifts some of the plasticity, but when the tonic is gone the phasic can’t do much about it.”
In new work, the lab is now looking at differences in gene expression between the two neurons to identify which proteins are responsible for the unique properties and plasticity of the tonic and phasic neurons. By defining the genetic underpinnings of their unique properties, the lab hopes to begin to get a handle on the molecular underpinnings of neuronal diversity in the brain.
In addition to Aponte-Santiago and Littleton, the paper’s other authors are postdoc Kiel Ormerod and Research Scientist Yulia Akbergenova, both of the Picower Institute and MIT. The National Institutes of Health and the JPB Foundation supported the study.