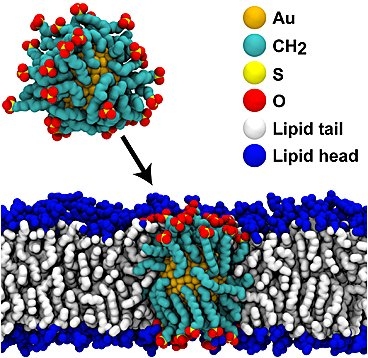
Cells are very good at protecting their precious contents — and as a result, it’s very difficult to penetrate their membrane walls to deliver drugs, nutrients or biosensors without damaging or destroying the cell. One effective way of doing so, discovered in 2008, is to use nanoparticles of pure gold, coated with a thin layer of a special polymer. But nobody knew exactly why this combination worked so well, or how it made it through the cell wall.
Now, researchers at MIT and the Ecole Polytechnique de Lausanne in Switzerland have figured out how the process works, and the limits on the sizes of particles that can be used. Their analysis appears in the journal Nano Letters, in a paper by graduate students Reid Van Lehn, Prabhani Atukorale, Yu-Sang Yang and Randy Carney and professors Alfredo Alexander-Katz, Darrell Irvine and Francesco Stellacci.
Until now, says Van Lehn, the paper’s lead author, “the mechanism was unknown. … In this work, we wanted to simplify the process and understand the forces” that allow gold nanoparticles to penetrate cell walls without permanently damaging the membranes or rupturing the cells. The researchers did so through a combination of lab experiments and computer simulations.
The team demonstrated that the crucial first step in the process is for coated gold nanoparticles to fuse with the lipids — a category of natural fats, waxes and vitamins — that form the cell wall. The scientists also demonstrated an upper limit on the size of such particles that can penetrate the cell wall — a limit that depends on the composition of the particle’s coating.
The coating applied to the gold particles consists of a mix of hydrophobic and hydrophilic components that form a monolayer — a layer just one molecule thick — on the particle’s surface. Any of several different compounds can be used, the researchers explain.
“Cells tend to engulf things on the surface,” says Alexander-Katz, an associate professor of materials science and engineering at MIT, but it’s “very unusual” for materials to cross that membrane into the cell’s interior without causing major damage. Irvine and Stellacci demonstrated in 2008 that monolayer-coated gold nanoparticles could do so; they have since been working to better understand why and how that works.
Since the nanoparticles themselves are completely coated, the fact that they are made of gold doesn’t have any direct effect, except that gold nanoparticles are an easily prepared model system, the researchers say. However, there is some evidence that the gold particles have therapeutic properties, which could be a side benefit.
Gold particles are also very good at capturing X-rays — so if they could be made to penetrate cancer cells, and were then heated by a beam of X-rays, they could destroy those cells from within. “So the fact that it’s gold may be useful,” says Irvine, a professor of materials science and engineering and biological engineering and member of the
Koch Institute for Integrative Cancer Research.
Significantly, the mechanism that allows the nanoparticles to pass through the membrane seems also to seal the opening as soon as the particle has passed. “They would go through without allowing even small molecules to leak through behind them,” Van Lehn says.
Irvine says that his lab is also interested in harnessing this cell-penetrating mechanism as a way of delivering drugs to the cell’s interior, by binding them to the surface coating material. One important step in making that a useful process, he says, is finding ways to allow the nanoparticle coatings to be selective about what types of cells they attach to. “If it’s all cells, that’s not very useful,” he says, but if the coatings can be targeted to a particular cell type that is the target of a drug, that could be a significant benefit.
Another potential application of this work could be in attaching or inserting biosensing molecules on or into certain cells, Van Lehn says. In this way, scientists could detect or monitor specific biochemical markers, such as proteins that indicate the onset or decline of a disease or a metabolic process.
In general, attachment to nanoparticles’ surface coatings could provide a key to cells’ interiors for “molecules that normally wouldn’t have any ability to get through the cell membrane,” Irvine says.
Vince Rotello, a professor of chemistry at the University of Massachusetts at Amherst who was not involved in this research, says this work is “careful, well thought out and elegantly presented.” He adds, “This study provides a very interesting alternative mechanism to cell uptake of nanomaterials that could open up new therapeutic pathways.”
The work was supported by the National Science Foundation, the National Cancer Institute and the U.S. Army Research Office.
Now, researchers at MIT and the Ecole Polytechnique de Lausanne in Switzerland have figured out how the process works, and the limits on the sizes of particles that can be used. Their analysis appears in the journal Nano Letters, in a paper by graduate students Reid Van Lehn, Prabhani Atukorale, Yu-Sang Yang and Randy Carney and professors Alfredo Alexander-Katz, Darrell Irvine and Francesco Stellacci.
Until now, says Van Lehn, the paper’s lead author, “the mechanism was unknown. … In this work, we wanted to simplify the process and understand the forces” that allow gold nanoparticles to penetrate cell walls without permanently damaging the membranes or rupturing the cells. The researchers did so through a combination of lab experiments and computer simulations.
The team demonstrated that the crucial first step in the process is for coated gold nanoparticles to fuse with the lipids — a category of natural fats, waxes and vitamins — that form the cell wall. The scientists also demonstrated an upper limit on the size of such particles that can penetrate the cell wall — a limit that depends on the composition of the particle’s coating.
The coating applied to the gold particles consists of a mix of hydrophobic and hydrophilic components that form a monolayer — a layer just one molecule thick — on the particle’s surface. Any of several different compounds can be used, the researchers explain.
“Cells tend to engulf things on the surface,” says Alexander-Katz, an associate professor of materials science and engineering at MIT, but it’s “very unusual” for materials to cross that membrane into the cell’s interior without causing major damage. Irvine and Stellacci demonstrated in 2008 that monolayer-coated gold nanoparticles could do so; they have since been working to better understand why and how that works.
Since the nanoparticles themselves are completely coated, the fact that they are made of gold doesn’t have any direct effect, except that gold nanoparticles are an easily prepared model system, the researchers say. However, there is some evidence that the gold particles have therapeutic properties, which could be a side benefit.
Gold particles are also very good at capturing X-rays — so if they could be made to penetrate cancer cells, and were then heated by a beam of X-rays, they could destroy those cells from within. “So the fact that it’s gold may be useful,” says Irvine, a professor of materials science and engineering and biological engineering and member of the
Koch Institute for Integrative Cancer Research.
Significantly, the mechanism that allows the nanoparticles to pass through the membrane seems also to seal the opening as soon as the particle has passed. “They would go through without allowing even small molecules to leak through behind them,” Van Lehn says.
Irvine says that his lab is also interested in harnessing this cell-penetrating mechanism as a way of delivering drugs to the cell’s interior, by binding them to the surface coating material. One important step in making that a useful process, he says, is finding ways to allow the nanoparticle coatings to be selective about what types of cells they attach to. “If it’s all cells, that’s not very useful,” he says, but if the coatings can be targeted to a particular cell type that is the target of a drug, that could be a significant benefit.
Another potential application of this work could be in attaching or inserting biosensing molecules on or into certain cells, Van Lehn says. In this way, scientists could detect or monitor specific biochemical markers, such as proteins that indicate the onset or decline of a disease or a metabolic process.
In general, attachment to nanoparticles’ surface coatings could provide a key to cells’ interiors for “molecules that normally wouldn’t have any ability to get through the cell membrane,” Irvine says.
Vince Rotello, a professor of chemistry at the University of Massachusetts at Amherst who was not involved in this research, says this work is “careful, well thought out and elegantly presented.” He adds, “This study provides a very interesting alternative mechanism to cell uptake of nanomaterials that could open up new therapeutic pathways.”
The work was supported by the National Science Foundation, the National Cancer Institute and the U.S. Army Research Office.