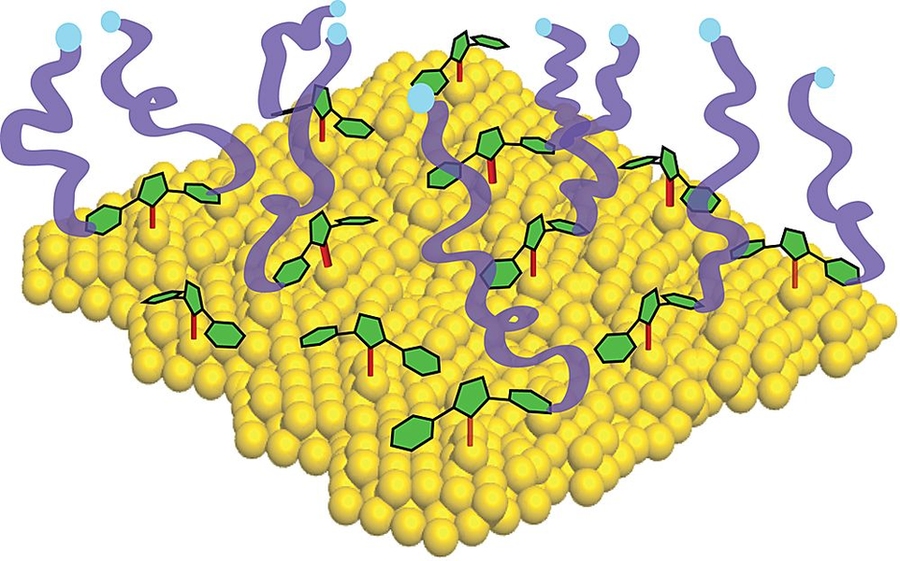
Over the past three decades, researchers have found various applications of a method for attaching molecules to gold; the approach uses chemicals called thiols to bind the materials together. But while this technique has led to useful devices for electronics, sensing and nanotechnology, it has limitations. Now, an MIT team has found a new material that could overcome many of these limitations.
The new approach uses a family of chemicals called carbenes to attach other substances to gold — and potentially to other material surfaces as well. The work, led by assistant professor of chemistry Jeremiah Johnson, has been published in the Journal of the American Chemical Society by a team that also included professor of chemistry Troy Van Voorhis and graduate students Aleksandr Zhukhovitskiy and Michael Mavros.
Thiols have two main limitations in binding other materials to gold, Johnson explains: The binding is relatively weak, so the attached molecules can come loose with heating, and the connection does not typically conduct electricity well, limiting use in electronic devices.
The MIT team envisioned that certain carbenes can overcome both hurdles. In so doing, they would enable a wide variety of applications, Johnson says, such as in the development of molecular electronics.
“You could scale electronic components down to the molecular level by wiring a molecule between two electrodes,” he says. “It would be the smallest possible component.”
Others have tried to do this with thiol-based connections, but these junctions have a rather large resistance. By comparison, preliminary indications suggest that carbenes could provide highly conductive linkages: “Electrons could flow through it like a wire,” Johnson says.
These carbenes could function as “surface anchors” to link many compounds to many different surface materials — a process known to chemists as “functionalizing” the surface. Johnson says, “I can count on one hand the number of methods you can use to functionalize surfaces, and they are different for different surfaces. If we could find a general one, that could make a big difference.”
Carbenes — specifically, a type the MIT team calls addressable N-heterocyclic carbenes (ANHCs) — may provide such a generalized solution. While further experiments will be needed to confirm the material’s performance, the technology holds much promise.
It was already known, Johnson says, that some carbenes can bond securely to a variety of metal surfaces, as well as many other materials. But there had been no investigation of their possible use as anchors, stably binding dissimilar materials.
Such combinations could be used as biosensors, for example: A molecule designed to bond with a specific biological marker could be attached to a gold wire, activating a circuit when that marker bonds with it. It could also be used to create protective surface coatings: antifouling surfaces to prevent buildup of biological deposits, or antibiotic coatings to prevent the spread of infections.
Another possible application might be to coat gold nanoparticles with a biomolecule that binds to tumors. The particles could then be heated using infrared light, killing the tumors with heat. ANHC coated surfaces could be beneficial in this regard, as they should be stable at higher temperatures, which would prevent particle degradation.
Once specific applications are found, the material has great potential because “it’s cheap to make, and you can make it at large scale,” Johnson says.
Brent Segal, chief scientist for nanotechnology at Lockheed Martin’s Advanced Technology Center in Billerica, Mass., says this work “represents a shift in the paradigm for attachment chemistry to metal atoms.” He adds that, “When I first learned of the approach, I said, ‘Finally!’ It seems as if someone might have come across something like this previously, but in terms of novelty, Johnson and his team have really executed beautifully to work out details for a viable system approach.”
Segal cautions, however, “As is often the case, this is still quite early-stage work and will have various hurdles yet to overcome. Scaling up the material and understanding the limitations of performance in various solutions … will be paramount.”
The work received funding from MIT’s Deshpande Center for Technological Innovation, the MIT Lincoln Laboratory, and the U.S. Department of Defense.
The new approach uses a family of chemicals called carbenes to attach other substances to gold — and potentially to other material surfaces as well. The work, led by assistant professor of chemistry Jeremiah Johnson, has been published in the Journal of the American Chemical Society by a team that also included professor of chemistry Troy Van Voorhis and graduate students Aleksandr Zhukhovitskiy and Michael Mavros.
Thiols have two main limitations in binding other materials to gold, Johnson explains: The binding is relatively weak, so the attached molecules can come loose with heating, and the connection does not typically conduct electricity well, limiting use in electronic devices.
The MIT team envisioned that certain carbenes can overcome both hurdles. In so doing, they would enable a wide variety of applications, Johnson says, such as in the development of molecular electronics.
“You could scale electronic components down to the molecular level by wiring a molecule between two electrodes,” he says. “It would be the smallest possible component.”
Others have tried to do this with thiol-based connections, but these junctions have a rather large resistance. By comparison, preliminary indications suggest that carbenes could provide highly conductive linkages: “Electrons could flow through it like a wire,” Johnson says.
These carbenes could function as “surface anchors” to link many compounds to many different surface materials — a process known to chemists as “functionalizing” the surface. Johnson says, “I can count on one hand the number of methods you can use to functionalize surfaces, and they are different for different surfaces. If we could find a general one, that could make a big difference.”
Carbenes — specifically, a type the MIT team calls addressable N-heterocyclic carbenes (ANHCs) — may provide such a generalized solution. While further experiments will be needed to confirm the material’s performance, the technology holds much promise.
It was already known, Johnson says, that some carbenes can bond securely to a variety of metal surfaces, as well as many other materials. But there had been no investigation of their possible use as anchors, stably binding dissimilar materials.
Such combinations could be used as biosensors, for example: A molecule designed to bond with a specific biological marker could be attached to a gold wire, activating a circuit when that marker bonds with it. It could also be used to create protective surface coatings: antifouling surfaces to prevent buildup of biological deposits, or antibiotic coatings to prevent the spread of infections.
Another possible application might be to coat gold nanoparticles with a biomolecule that binds to tumors. The particles could then be heated using infrared light, killing the tumors with heat. ANHC coated surfaces could be beneficial in this regard, as they should be stable at higher temperatures, which would prevent particle degradation.
Once specific applications are found, the material has great potential because “it’s cheap to make, and you can make it at large scale,” Johnson says.
Brent Segal, chief scientist for nanotechnology at Lockheed Martin’s Advanced Technology Center in Billerica, Mass., says this work “represents a shift in the paradigm for attachment chemistry to metal atoms.” He adds that, “When I first learned of the approach, I said, ‘Finally!’ It seems as if someone might have come across something like this previously, but in terms of novelty, Johnson and his team have really executed beautifully to work out details for a viable system approach.”
Segal cautions, however, “As is often the case, this is still quite early-stage work and will have various hurdles yet to overcome. Scaling up the material and understanding the limitations of performance in various solutions … will be paramount.”
The work received funding from MIT’s Deshpande Center for Technological Innovation, the MIT Lincoln Laboratory, and the U.S. Department of Defense.