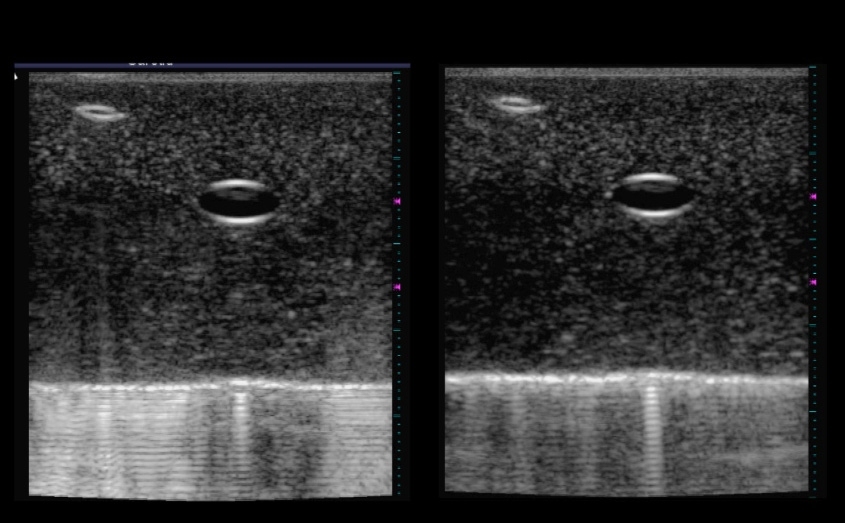
Ultrasound images, known as sonograms, have become a familiar part of pregnancy, allowing expectant parents a view of their unborn child. But new research at MIT could improve the ability of untrained workers to perform basic ultrasound tests, while allowing trained workers to much more accurately track the development of medical conditions, such as the growth of a tumor or the buildup of plaque in arteries.
The improvements to this widely used technology could provide detailed information far beyond what is possible with existing systems, the researchers say. The work, led by Brian W. Anthony, co-director of MIT’s Medical Electronic Device Realization Center (MEDRC) and director of the Master of Engineering in Manufacturing Program, was recently presented at the International Symposium on Biomedical Imaging in Barcelona, Spain.
There are two key elements to the improvements engineered by Anthony and his team. First, the researchers devised a way to adjust for variations in the force exerted by a sonographer, producing more consistent images that can compensate for body motions such as breathing and heartbeat. Second, they provided a way to map the exact location on the skin where one reading was taken, so that it can be precisely matched with later readings to detect changes in the size or location of a tumor, clot or other structure.
Together, the two improvements could make sonography a much more precise tool for monitoring the progression of disease, Anthony says. The devices are currently undergoing three clinical trials, including one at Boston Children’s Hospital focused on monitoring the progression of patients with Duchenne Muscular Dystrophy (DMD).
In that trial, Anthony says, researchers are trying to determine “how fast the muscle deteriorates, and how effective different medications are.” It’s important to have a reliable way of monitoring changes in muscle, he says. The study is aimed at determining whether ultrasound analysis can serve as a convenient, noninvasive, clinically meaningful way of monitoring disease progression in DMD.
The new device maintains constant force through the addition of a force sensor to its probe tip and servomotors that can respond almost instantly to changes in force. That, in turn, makes it possible to analyze how the image varies as the force increases, which can provide important diagnostic information about the elasticity of skin, muscle and other tissues.
To provide accurate positioning, a tiny camera and lens mounted on the probe can reveal skin patterns that are distinctive and constant, similar to fingerprints. “Skin patterns are pretty unique,” Anthony says; his team’s system, using software to compare new images with earlier ones, “can get you back to that same patch of skin,” something that is impossible to do manually.
Anthony likens that precise positioning to “an on-the-patient GPS system” for locating structures in the body. The ability to take images over time from exactly the same position makes it possible to monitor changing tissues quite precisely: The imaging system can determine the volume of a near-surface tumor or other feature to within an accuracy of 1 to 2 percent, he says. There are existing ways to get this kind of accuracy, but these require expensive specialized equipment that few hospitals have.
Besides the potential for these advanced diagnostic capabilities, enhanced control over testing could make it possible for relatively untrained health care workers to administer basic ultrasound pregnancy tests — especially in remote, underserved areas where trained sonographers may not be available. The various control techniques “take the uncertainty out” of the process, Anthony says.
Craig Steiner, an anesthesiologist at Chester County Hospital in Pennsylvania, says, “I’m excited about the prospects” of these improved systems. “The reproducibility of the scan with consistent pressure and picture quality would help with remote readings of locally done scans. This could be relevant for teleradiology, which is an area ripe for expansion.”
Steiner adds: “The field of ultrasound is still developing. Ultrasound will partially replace CT scans, reduce radiation exposure to patients and make diagnosing easier when away from the high-cost hospitals. It can help our world provide care at a more reasonable cost with a new paradigm of care.”
The research was conducted by Anthony, mechanical engineering graduate student Matthew Gilbertson and electrical engineering graduate student Shih-Yu Sun. The work has been supported by the Singapore-MIT Alliance, Terason and GE Healthcare.