A team including MIT researchers has demonstrated for the first time that artificially created stem cells can be used to treat symptoms of Parkinson's disease in rats.
The work, reported in the April 7 online Early Edition of the Proceedings of the National Academy of Sciences, could lead to successful treatments for human patients of Parkinson's, the degenerative neurological disorder. However, the researchers pointed out that hurdles associated with reprogramming cells must first be cleared.
Last year, U.S. and Japanese researchers reported that they had reprogrammed normal human skin cells into behaving like embryonic stem cells, which can create almost any cell in the body. In December, a team from the Whitehead Institute of Biomedical Research found that the reprogrammed skin cells, known as induced pluripotent stem (IPS) cells, could be used to treat mice with a human sickle-cell anemia disease trait.
The latest discovery involved members of the same Whitehead lab. They found that nerve cells derived from the reprogrammed cells can be successfully integrated in animal brains and improve symptoms of a neurodegenerative condition similar to Parkinson's.
"This is the first demonstration that reprogrammed cells can integrate into the neural system or positively affect neurodegenerative disease," said Marius Wernig, lead author of the article and a postdoctoral researcher in the laboratory of Rudolf Jaenisch, a Whitehead Member and a professor of biology at MIT.
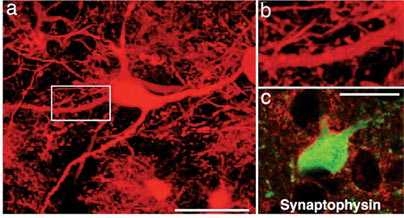
In one experiment, Wernig transplanted neural precursor cells into brain cavities of mouse embryos. The mice were naturally delivered and analyzed nine weeks after the transplantation. Wernig found that transplanted cells formed clusters where they had been injected and then migrated extensively into the surrounding brain tissues. Using electrophysiological studies conducted by Jian-Ping Zhao, a researcher in Martha Constantine-Paton's laboratory from MIT's McGovern Institute for Brain Research, and structural analysis, Wernig also saw that the neural precursor cells that migrated had differentiated into several subtypes of neural cells, including neurons and glia, and had functionally integrated into the brain.
In another study, Wernig used a rat model for Parkinson's disease, which is caused by insufficient levels of the hormone dopamine in a specific part of the midbrain. To mimic this state, the dopamine-producing neurons were killed on one side of the rat brains. In collaboration with Ole Isacson's group at McLean Hospital/ Harvard Medical School, Wernig then grafted differentiated dopamine neurons into a part of the rat brains called the striatum.
Four weeks after surgery, the rats were tested for dopamine-related behavior. In response to amphetamine injections, rats typically walk in circles toward the side with less dopamine activity in the brain. Eight of nine rats that received the dopamine neuron transplants showed markedly less or even no circling. Eight weeks after transplantation, the researchers could see that the dopamine neurons had extended into the surrounding brain.
"This experiment shows that in vitro reprogrammed cells can in principle be used to treat Parkinson's disease," said Jaenisch. "It's a proof of principle experiment that argues, yes, these cells may have the therapeutic promise that people ascribe to them."
Jaenisch and Wernig are optimistic that this work could be applied to human patients, but they cautioned that major challenges remain. Those include finding alternatives to the potentially cancer-causing retroviruses used to transform the skin cells into IPS cells and figuring out the best methods and places to transplant the neural precursor cells in humans.
The research was supported by the Ellison Medical Foundation and the National Institutes of Health.
A version of this article appeared in MIT Tech Talk on April 9, 2008 (download PDF).