Hepatitis C, a virus that can cause liver failure or cancer, infects about 200 million people worldwide. Existing drugs are not always effective, so many patients end up on long liver-transplant waiting lists.
One reason that no better treatment options exist is the lack of a suitable liver tissue model to test new drugs in the laboratory. But now, researchers from MIT and Rockefeller University have successfully grown hepatitis C viruses in otherwise healthy liver cells.
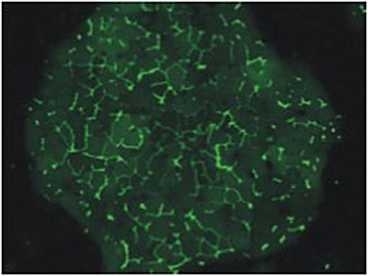
In the new tissue model, liver cells are precisely arranged on a specially patterned plate and surrounded by supportive cells, allowing them to live and function for four to six weeks. The cells can be infected with hepatitis C for two to three weeks, giving researchers the chance to study the cells’ responses to different drugs.
“With this model system, one can study hepatitis C and its chronic effects in greater mechanistic detail,” says Salman Khetani, former MIT postdoctoral associate and an author of two recent papers on the work. “Since it uses normal human liver cells rather than cancer-derived ones, our system may provide a better understanding of how hepatitis C progresses in humans, and of potential cures.”
The research team, led by Sangeeta Bhatia, professor in the Harvard-MIT Division of Health Sciences and Technology and MIT’s Department of Electrical Engineering and Computer Science, and Charles Rice of Rockefeller University, reported the new method in recent papers published in the Proceedings of the National Academy of Sciences and Nature Biotechnology.
Patterning cells
Previously, researchers have been able to induce cancerous liver cells to survive and reproduce outside the body, but those cells are not sufficient for studying hepatitis C because their responses to infection are different from those of normal liver cells.
To create their new model, researchers used healthy liver cells that had been cryogenically preserved and grew them on special plates with micropatterns that direct the cells where to grow. The liver cells were strategically interspersed with other cells called fibroblasts that support the growth of liver tissue.
“If you just put cells on a surface in an unorganized way, they lose their function very quickly,” says Bhatia, who is also a Howard Hughes Medical Institute Investigator. “If you specify which cells sit next to each other, you can extend the lifetime of the cells and help them maintain their function.”
Not only did the researchers successfully grow cells infected with hepatitis C, they also developed new ways to confirm that the viruses have entered cells and replicated themselves. Those techniques, reported in the Jan. 31 online edition of Nature Biotechnology, could allow drug developers to more easily test potential new drugs.
To test whether viruses have entered cells, the researchers engineered hepatitis C-like viral particles to express a fluorescent protein that glows when the virus goes into cells. That could help researchers test potential drugs that block viral entry into cells, by allowing them to see whether the virus has made it into the cells or not.
They also developed a test that reveals whether hepatitis C viruses have successfully reproduced inside an infected cell. Such a test would help researchers test the efficiency of potential drugs that block viral replication.
In these studies, the researchers used a strain of hepatitis C isolated from a Japanese patient with severe hepatitis, the only strain ever successfully grown in the lab. They hope to modify the system so they can grow additional strains, including those commonly found in North America, which would allow for more thorough drug testing.
Raymond Chung, director of hepatology at Massachusetts General Hospital, says the new model represents a significant improvement over existing tissue models, derived from liver tumors. With this new technology, researchers should be able to get a more accurate picture of virus-host interactions and the effectiveness of viral inhibitors, he says. They could also gain insight into why hepatitis C persists as a chronic infection in the majority of patients. “This model allows us to really get at these issues in a more bona fide way, using real viruses and, for the first time, real liver cells,” he says.
One reason that no better treatment options exist is the lack of a suitable liver tissue model to test new drugs in the laboratory. But now, researchers from MIT and Rockefeller University have successfully grown hepatitis C viruses in otherwise healthy liver cells.
In the new tissue model, liver cells are precisely arranged on a specially patterned plate and surrounded by supportive cells, allowing them to live and function for four to six weeks. The cells can be infected with hepatitis C for two to three weeks, giving researchers the chance to study the cells’ responses to different drugs.
“With this model system, one can study hepatitis C and its chronic effects in greater mechanistic detail,” says Salman Khetani, former MIT postdoctoral associate and an author of two recent papers on the work. “Since it uses normal human liver cells rather than cancer-derived ones, our system may provide a better understanding of how hepatitis C progresses in humans, and of potential cures.”
The research team, led by Sangeeta Bhatia, professor in the Harvard-MIT Division of Health Sciences and Technology and MIT’s Department of Electrical Engineering and Computer Science, and Charles Rice of Rockefeller University, reported the new method in recent papers published in the Proceedings of the National Academy of Sciences and Nature Biotechnology.
Patterning cells
Previously, researchers have been able to induce cancerous liver cells to survive and reproduce outside the body, but those cells are not sufficient for studying hepatitis C because their responses to infection are different from those of normal liver cells.
To create their new model, researchers used healthy liver cells that had been cryogenically preserved and grew them on special plates with micropatterns that direct the cells where to grow. The liver cells were strategically interspersed with other cells called fibroblasts that support the growth of liver tissue.
“If you just put cells on a surface in an unorganized way, they lose their function very quickly,” says Bhatia, who is also a Howard Hughes Medical Institute Investigator. “If you specify which cells sit next to each other, you can extend the lifetime of the cells and help them maintain their function.”
Not only did the researchers successfully grow cells infected with hepatitis C, they also developed new ways to confirm that the viruses have entered cells and replicated themselves. Those techniques, reported in the Jan. 31 online edition of Nature Biotechnology, could allow drug developers to more easily test potential new drugs.
To test whether viruses have entered cells, the researchers engineered hepatitis C-like viral particles to express a fluorescent protein that glows when the virus goes into cells. That could help researchers test potential drugs that block viral entry into cells, by allowing them to see whether the virus has made it into the cells or not.
They also developed a test that reveals whether hepatitis C viruses have successfully reproduced inside an infected cell. Such a test would help researchers test the efficiency of potential drugs that block viral replication.
In these studies, the researchers used a strain of hepatitis C isolated from a Japanese patient with severe hepatitis, the only strain ever successfully grown in the lab. They hope to modify the system so they can grow additional strains, including those commonly found in North America, which would allow for more thorough drug testing.
Raymond Chung, director of hepatology at Massachusetts General Hospital, says the new model represents a significant improvement over existing tissue models, derived from liver tumors. With this new technology, researchers should be able to get a more accurate picture of virus-host interactions and the effectiveness of viral inhibitors, he says. They could also gain insight into why hepatitis C persists as a chronic infection in the majority of patients. “This model allows us to really get at these issues in a more bona fide way, using real viruses and, for the first time, real liver cells,” he says.