With the annual flu season upon us, vaccines remain the primary weapons for fighting influenza outbreaks. Yet, there frequently aren't enough doses to go around.
One major reason is that the contracts between governments and manufacturers do not provide vaccine makers with enough of a financial incentive to produce sufficient doses. Now, MIT researchers and colleagues have come up with a new model for contracts they hope will benefit both parties and prevent vaccine shortages.
The work is the subject of a paper that was recently awarded the 2006 Pierskalla Best Paper Award in health care management science by the Institute for Operations Research and the Management Sciences (INFORMS). The paper, "Supply Chain Coordination and Influenza Vaccination," is under review for publication.
The authors are Professor David Simchi-Levi of MIT's Engineering Systems Division and Department of Civil and Environmental Engineering, who was recently named an INFORMS fellow; Hamed Mamani, a doctoral student at the MIT Operations Research Center; and Stephen E. Chick, associate professor of technology management at INSEAD, one of the world's largest graduate business schools, with campuses in Singapore and France.
"Manufacturers and governments have conflicting objectives," said Simchi-Levi. "The manufacturers' objective is to reduce risk, maximize profit and control production so they are not stuck with an excess supply (of vaccine), while governments and health care organizations need to balance the vaccination program's cost with the public health benefit of not spreading the disease," he said.
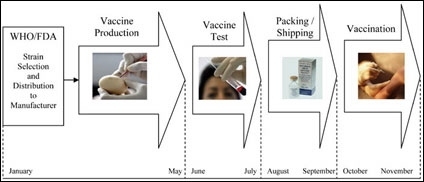
A new type of contract is needed because manufacturing and distributing flu vaccines brings a unique set of problems. New flu vaccines must be made in a relatively short period, only six months. Public health officials must first identify which strains are most likely to cause sickness, and then the manufacturers must make, test and distribute the doses.
The usual annual vaccine protects against three strains of flu. Strains of flu experience slight mutations to their genome, a phenomenon known as antigenic drift. Because of this, the immunity acquired for one year's vaccine may not combat the next year's strains of flu.
Thus, a major challenge is that the strains of flu virus change every year, so old vaccine inventory must be discarded and new vaccine made. Another challenge is that because of the biological nature of the production process, the amount of vaccine produced successfully varies greatly.
It is also difficult to determine the exact number of high-risk patients, such as children and the elderly, who will need vaccine. That is a disincentive for manufacturers to make enough doses to cover a worst-case scenario; they will lose money on any excess.
Simchi-Levi and colleagues have devised a model in which the government and manufacturer would share the costs associated with possible overproduction. That should provide manufacturers with an incentive to make more vaccines, even if they are not sure all the doses will be used.
"There also is an incentive for the government to pay for the extra doses," Simchi-Levi said. If influenza spreads, the "social costs," such as hospitalization and lost wages, range from $1 million to $6 million per 100,000 people in industrialized countries, according to the World Health Organization. Even with vaccines, influenza kills up to 500,000 people each year.
Simchi-Levi said the importance of the new model is that it explains why there aren't enough vaccines and suggests a way for governments and manufacturers to mitigate the problem through financial risk-sharing.
A version of this article appeared in MIT Tech Talk on January 10, 2007 (download PDF).