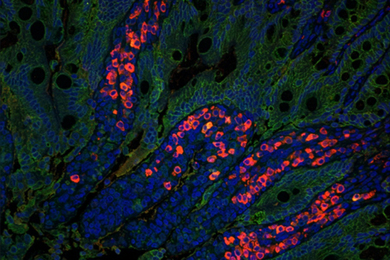
CAMBRIDGE, Mass. -- A defect in the modification process of messenger RNA may play a role in a common malignant brain tumor, Massachusetts Institute of Technology researchers will report in the Nov. 20 online edition of Proceedings of the National Academy of Sciences.
This study -- the first evidence for a molecular link between changes in RNA editing and human disease -- may also help explain the occurrence of epileptic seizures in association with the tumor.
RNA editing enables cells to add, delete or modify bases-the building blocks of genetic material-to create new "recipes" for assembling different proteins.
There are different types of RNA editing. MIT biologists Stefan Maas and Alexander Rich, with colleagues from Friedrich Schiller University in Germany, are studying base substitution, which gives rise to a single amino acid change in the resulting protein.
In this study, the researchers looked at whether the cellular machinery for substitution RNA editing might be associated with the progression of cancerous growth within the human brain, where a number of important targets for RNA editing reside. "We asked the question, 'Is it possible that glioma, a common malignant brain tumor that often causes epileptic seizures, could be related to modification of the RNA editing system?'" Rich said.
NATURE'S TRICKS
Just as 26 letters of the alphabet form a multitude of words, our genetic material creates many different configurations of four basic molecules. The substitution of one letter can make a big difference in the meaning of a word (such as "dancer" or "cancer"), and editing one base in a strip of RNA can produce very different results when it is later translated into protein.
"It's nature's way of creating functional diversity," Rich said. "You start with one function, and with a little bit of tinkering, you create another function." In this manner, increasingly complex substances and organisms can be made out of a limited number of genes, the universal material of life.
The growth of tumors starting with early-stage cancer and all the way through the most malignant and invasive growth involves a multitude of cellular changes. Any gene product involved in signalling pathways within the cell that affect cell growth, survival and proliferation are possible suspects in initiating or promoting cancerous growth.
A mistake in RNA editing is one possible culprit.
A DEADLY TUMOR
The most malignant common tumor of the central nervous system is glioblastoma multiforme (GBM), which is almost always fatal. Patients with malignant gliomas tend to have epileptic seizures.
By looking at the level of editing present in samples of human glioma tissues, the researchers found a lot of unedited messenger RNA. "This is a somewhat surprising finding," Rich said. And although it is not known what relevance the editing mechanism has to tumor formation, "it does raise the possibility that symptoms of gliomas may be related to this." This is supported by results from mouse models. Genetically altered mice suffer severe epileptic seizures and premature death when the editing process is compromised.
Normally, virtually all of a particular RNA is edited, or changed, and this seems to be essential for normal brain function. The protein product of the edited RNA regulates the influx of ions into the cell. The unedited RNA results in a protein with profoundly altered ion gating properties. This effects the way brain cells communicate with their neighboring cells. A malfunction in RNA editing could cause an imbalance that would lead to seizures and might modify tumor growth.
Rich says that with increasing knowledge of the recently discovered phenomena of RNA editing, researchers may one day be able to help relieve symptoms such as seizures related to gliomas, even if they are not able to reverse the tumor itself.
"Currently, together with undergraduate researcher Jay Shrestha, an MIT junior biology major, we are examining the possible link between the malignancy of a tumor and the extent by which RNA editing is depressed," said Maas, lead author of the study. Such a link would make the RNA editing status a valuable marker for the categorization and prognosis of gliomas.
"When we get to understand the system, we may be able to learn how to control, regulate and modulate it," Rich said.
Human glioblastoma tissue samples were provided by the Institute of Pathology of the Friedrich Schiller University and Massachusetts General Hospital.
The work is funded by the Anna Fuller Fund for Molecular Oncology, the National Institutes of Health, the National Science Foundation and the National Foundation for Cancer Research.
Copies of the article are now available to reporters from the PNAS news office. Call (202) 334-2138 or email pnasnews@nas.edu.